Chemical Name:Melanotan II (MT-II)
CAS:121062-08-6
Synonyms:Ac-Nle-cyclo[Asp-His-D-Phe-Arg-Trp-Lys]-NH₂, Melanotan 2, MT-2
Molecular Formula:C₅₀H₆₈N₁₄O₁₀
Molecular Weight:1024.2 g/mol
Appearance:White to off-white powder
Solubility:Soluble in water (>10 mg/mL), slightly soluble in organic solvents (e.g., DMSO)
Stability:Stable at room temperature when lyophilized; aqueous solutions may degrade over time (store at -20°C)
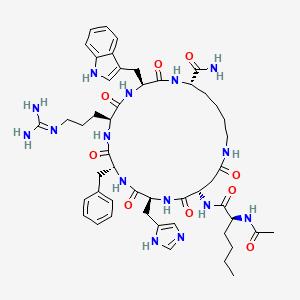
Peptide Sequence Ac-Nle-c[Asp-His-D-Phe-Arg-Trp-Lys]-NH₂ (Cyclic heptapeptide with a lactam bridge between Asp and Lys)
Biological Function:Synthetic analog of α-MSH (melanocyte-stimulating hormone); non-selective agonist of melanocortin receptors (MC1R, MC3R, MC4R, MC5R). Promotes melanogenesis, libido enhancement, and potential appetite suppression.
Primary Uses:Research compound (originally studied for sunless tanning, erectile dysfunction, and weight loss). Not FDA-approved; sold as an unregulated peptide.
Half-Life ~2–3 hours (subcutaneous administration)
Side Effects:Nausea, facial flushing, appetite suppression, spontaneous erections (in males), darkening of existing moles/freckles.

| Parameter | Specification | Test Method | Acceptance Criteria | Results (Example) |
|---|
| Appearance | Visual inspection | Visual (USP/Ph. Eur.) | White to off-white, free-flowing powder | Complies |
| Identification | HPLC retention time vs. reference standard | HPLC (UV detection, 220 nm) | Matches reference standard (±2%) | 99.8% match |
| Purity (HPLC) | ≥ 98% (area percent) | RP-HPLC (C18 column, gradient elution) | ≥ 98.0% | 99.2% |
| Related Substances | Individual impurities ≤ 1.0%; Total impurities ≤ 2.0% | HPLC | Complies with ICH Q3 guidelines | Max impurity: 0.5% |
| Amino Acid Composition | Matches theoretical sequence (Nle, Asp, His, D-Phe, Arg, Trp, Lys) | Amino Acid Analysis (HPLC/MS) | ±10% of expected ratios | Complies |
| Water Content | ≤ 5.0% (w/w) | Karl Fischer titration | ≤ 5.0% | 3.2% |
| Residual Solvents | MeOH, DMSO, TFA ≤ ICH Q3C limits | GC-HS (Headspace Gas Chromatography) | MeOH ≤ 3000 ppm; DMSO ≤ 500 ppm | Not detected |
| Peptide Content | 95–105% of labeled amount | UV spectrophotometry (280 nm) | 95–105% | 101.5% |
| Microbial Limits | Total aerobic count ≤ 100 CFU/g; Absence of pathogens (E. coli, Salmonella, etc.) | USP <61>, <62> | Complies with USP/EP | Complies |
| Endotoxins | < 10 EU/mg | LAL test (Gel Clot Method) | < 10 EU/mg | < 5 EU/mg |
| pH (1% solution) | 5.0–7.0 | Potentiometry | 5.0–7.0 | 6.2 |
| Sterility (if applicable) | Sterile (for injectable formulations) | Membrane filtration (USP <71>) | No growth after 14 days | Sterile |

Structure: Cyclic peptide with a modified amino acid sequence (D-Phe enhances receptor binding).
Mechanism: Activates melanocortin receptors, stimulating melanin production and modulating sexual appetite/metabolism.
Legal Status: Unapproved for medical use; classified as a research chemical in most countries



 China
China