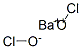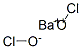Barium hypochlorite is a colorless, sand-like,crystalline solid, often used in solution. A strong oxidizer.There is not much information available on this compound.Reacts with water
Barium hypochlorite has the formula, BaClO2 and the
molecular weight of 240.234 g/mol. Its CAS number is
13477-10-6. It is white-colored crystalline solid that is
denser than water. Contact may irritate skin, eyes and
mucous membranes. It may be toxic by ingestion. It
will accelerate the burning of combustible materials.
Prolonged exposure to heat or flames may result in an
explosion. It reacts with acids, evolving chlorine, an irritating,
corrosive and toxic gas. It reacts fiercely with
cyanides when heated or by friction. It may form explosive
mixtures with combustible material, powdered
metals or ammonium compounds. It is not offered
commercially for sale nor is the scientific literature concerning
its uses or physical and chemical properties
readily available.
A white-colored crystalline solid. Denser than water. Contact may irritate skin, eyes and mucous membranes. May be toxic by ingestion. May accelerate the burning of combustible materials. Prolonged exposure to heat or flames may result in an explosion.
Salts of hypochlorous acid, HClO. Generally toxic, irritants and powerful oxidizers, particularly in the presence of water or at higher temperature as they decompose to release oxygen and chlorine gases. On contact with urea they form the highly explosive NCl3 . When heated or on contact with acids, they produce highly toxic fumes of chlorine gas [Sax, 9th ed., 1996, p. 1905].
Toxic by ingestion. Inhalation of dust is toxic. Fire may produce irritating, corrosive and/or toxic gases. Contact with substance may cause severe burns to skin and eyes. Runoff from fire control or dilution water may cause pollution.
These substances will accelerate burning when involved in a fire. May explode from heat or contamination. Some may burn rapidly. Some will react explosively with hydrocarbons (fuels). May ignite combustibles (wood, paper, oil, clothing, etc.). Containers may explode when heated. Runoff may create fire or explosion hazard.
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seekmedical attention immediately. If this chemical contactsthe skin, remove contaminated clothing and wash immediately with soap and water. Seek medical attention immediately. If this chemical has been inhaled, remove fromexposure, begin rescue breathing (using universal precautions, including resuscitation mask) if breathing hasstopped and CPR if heart action has stopped. Transferpromptly to a medical facility. When this chemical hasbeen swallowed, get medical attention. Give large quantities of water and induce vomiting. Do not make an unconscious person vomit.
Color Code—Yellow: Reactive Hazard; Store in alocation separate from other materials, especially flammables and combustibles. Store in tightly closed containersin a cool, well-ventilated area away from water and steam.Sources of ignition, such as smoking and open flames, areprohibited where Barium Hypochlorite is used, handled, orstored in a manner that could create a potential fire orexplosion hazard. Avoid any possible contact with theincompatible materials cited above. See OSHA Standard1910.104 and NFPA 43A Code for the Storage of Liquidand Solid Oxidizers for detailed handling and storageregulations.
Barium hypochlorite (with more than 22% available chlorine) requires an “OXIDIZER” label. It falls inHazard Class 5.1 and Packing Group II.
Barium hypochlorite can affect youwhen breathed in and may enter the body through the skin.Contact can irritate and even burn the eyes and skin.Breathing the dust or mist can irritate the nose, throat, andbronchial tubes, causing cough and phlegm. Exposure toeither form can affect the nervous system and cause hypo