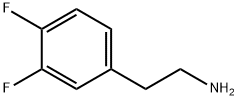
Synthesis of intermediate 2-(3,4-difluorophenyl)ethylamine (I-49b): the preparation and post-treatment steps were carried out with reference to the preparation of intermediate I-15b. The specific operation was as follows: E-1,2-difluoro-4-(2-nitrovinyl)benzene (I-49a, 5 g, 27.027 mmol) was dissolved in anhydrous THF (81 mL), and LiBH4 (2.35 g, 108.1081 mmol) and chlorotrimethylsilane (27.63 mL, 216.216 mmol) were added sequentially at 0 °C. The reaction mixture was stirred at room temperature for 3 days. Upon completion of the reaction, 4.2 g of the target product was obtained by post-processing in 100% yield. The product was characterized by 1H NMR (DMSO, 300 MHz): δ 8.7 (broad peak, 2H), 7.5-7.3 (multiple peaks, 2H), 7.2-7.0 (multiple peaks, 1H), 3.0-2.8 (multiple peaks, 4H).LCMS analysis showed a purity of 98.87%, m/z = 158.1 (M + 1).
![1,2-difluoro-4-[(E)-2-nitroethenyl]benzene](/CAS/20200401/GIF/1025692-47-0.gif)