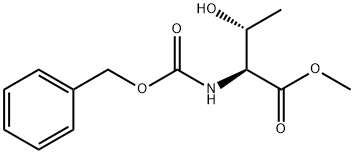
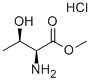
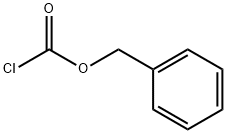
In a 2L beaker, 100 g of L-threonine methyl ester hydrochloride was dissolved in 600 ml of water and the pH was adjusted to a suitable range using sodium bicarbonate. Subsequently, 110 g of benzyl chloroformate (Z-Cl) was slowly added dropwise and the pH of the reaction system was kept between 8-9, which was adjusted using 2 M sodium hydroxide solution. After completion of the reaction, 1 L of ethyl acetate was added for extraction. The organic phase was washed sequentially with 500 ml of water and 500 ml of saturated saline, and then dried with 100 g of anhydrous sodium sulfate. After filtration, it was concentrated to dryness under reduced pressure to obtain a solid product. The solid was dissolved in 600 ml of a suitable solvent for crystallization, filtered and dried to give 140 g of N-benzyloxycarbonyl-L-threonine methyl ester (Z-Thr-OMe) in 89.1% yield.
[1] Patent: CN106631900, 2017, A. Location in patent: Paragraph 0015
[2] Journal of the Chemical Society - Perkin Transactions 1, 1999, # 24, p. 3697 - 3703
[3] Justus Liebigs Annalen der Chemie, 1962, vol. 656, p. 190 - 204
[4] Journal of Organic Chemistry, 1985, vol. 50, # 23, p. 4515 - 4523
[5] Patent: WO2010/17060, 2010, A1. Location in patent: Page/Page column 44-45