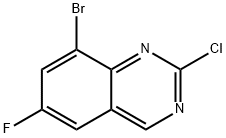
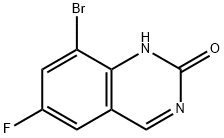
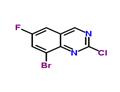
Step 5. Synthesis of 8-bromo-2-chloro-6-fluoroquinazoline: 0.5 M of 8-bromo-6-fluoroquinazolin-2(1H)-one was suspended in phosphorochloridic acid chloride and heated to 110 °C in an oil bath. During the reaction, the suspension transformed to a brown solution within 20 min. The reaction was completed after 1 h as monitored by LCMS. Subsequently, the excess phosphorus trichloride was removed by distillation under reduced pressure. The residue was carefully poured into ice water and the pH was adjusted with aqueous sodium bicarbonate to 7. The reaction mixture was extracted with ethyl acetate, the organic phases were combined, washed sequentially with water and saturated brine, dried over anhydrous sodium sulfate, and concentrated under reduced pressure to afford the target compound 8-bromo-2-chloro-6-fluoroquinazoline in 89% yield. The product was characterized by electrospray mass spectrometry (ES/MS) with m/z of 261/263 (MH+).