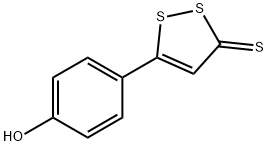
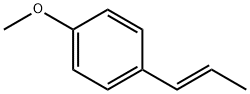
The general procedure for the synthesis of 5-(4-hydroxyphenyl)-3H-1,2-dithio-3-thione from anethole was as follows: anethole (1) (32.5 g, 0.21 mol) was mixed with sulfur powder (45 g, 1.40 mol) in N,N-dimethylformamide (250 mL) and the reaction was carried out by heating and refluxing for 8 hours. After completion of the reaction, the solvent was removed by distillation under reduced pressure and the resulting residue was dissolved in toluene. Subsequently, the toluene solution was extracted with 2N aqueous sodium hydroxide solution to give an orange solid precipitate (8.5 g). The solid product was dissolved in boiling water and hydrochloric acid was added slowly to acidity to precipitate an orange precipitate (2) in 50% yield with a melting point of 205 °C (literature value 188-189 °C). The structure of the product was confirmed by 1H NMR (DMSO-d6) and mass spectrometry (ESI): 1H NMR (DMSO-d6) δ 6.86 (d, 2H), 7.68 (s, 1H), 7.75 (d, 2H), 10.51 (s, 1H, -OH); MS (ESI) m/z 225 [M-H]-.