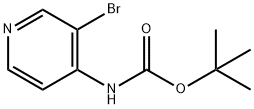
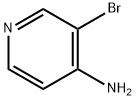
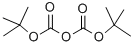
In a dry reaction flask, 4-amino-3-bromopyridine (10.0 g, 57.803 mmol) was dissolved in tetrahydrofuran (THF, 500 mL), and triethylamine (8.8 g, 86.705 mmol) and di-tert-butyl dicarbonate (Boc anhydride, 37.89 g, 173.410 mmol) were added sequentially under stirring. The reaction mixture was stirred continuously for 2 hours at room temperature. After completion of the reaction, the mixture was poured into water (700 mL) and extracted with ethyl acetate (EtOAc, 500 mL x 2). The organic phases were combined, dried over anhydrous sodium sulfate and concentrated under reduced pressure to give the crude product. The crude product was purified by column chromatography to afford tert-butyl (3-bromopyridin-4-yl)carbamate (37.0 g, 136.029 mmol) in a calculated yield based on 4-amino-3-bromopyridine. Mass spectrometry (MS) analysis showed a molecular ion peak of 274.95 (M + 2).
[1] Angewandte Chemie - International Edition, 2016, vol. 55, # 39, p. 11859 - 11862
[2] Angew. Chem., 2016, vol. 128, p. 12038 - 12041,4
[3] Patent: US2006/46983, 2006, A1. Location in patent: Page/Page column 186
[4] Patent: WO2018/69468, 2018, A1. Location in patent: Page/Page column 76