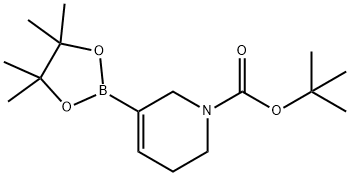
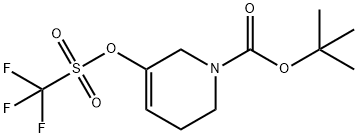
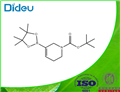
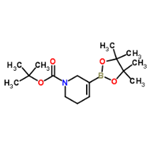
General procedure: PdCl2 (dppf) (0.16 g, 0.22 mmol), KOAc (2.18 g, 22.2 mmol), bis(pinacolato)diboron (2.07 g, 8.13 mmol), and dppf (0.12 g, 0.22 mmol) were added to a round-bottomed flask and the flask flushed with argon. A solution of degassed tert-butyl 5-(((trifluoromethyl)sulfonyl)oxy)-3,6-dihydropyridine-1(2H)-carboxylate (2.45 g, 7.40 mmol) in dioxane (70 mL) was added to the flask. The reaction mixture was heated to 80 °C and stirred for 16 hours. After completion of the reaction, it was filtered through a glass sintered funnel to remove the solid KOAc and the filtrate was concentrated under reduced pressure. Purification by silica gel column chromatography (eluent: 5% EtOAc in hexane solution) afforded tert-butyl 3-boronic acid pinacol esteryl-5,6-dihydropyridine-1(2H)-carboxylate (1.62 g, 71%) as a colorless oil.1H NMR (400 MHz, CDCl3): δ 6.69-6.60 (m, 1H), 3.98 (br s, 2H), 3.49-3.42 (m, 2H), 2.24-2.16 (m, 2H), 1.47 (s, 9H), 1.27 (s, 12H). LC-MS (ESI, m/z): calculated value C18H28BNO4 [M+H]+ 310.2, measured value 311.0.