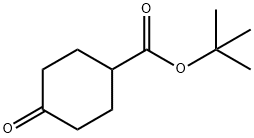
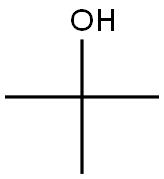
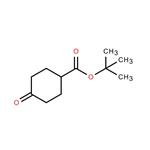
General procedure for the synthesis of tert-butyl 4-oxocyclohexanecarboxylate from 4-cyclohexanonecarboxylic acid and tert-butanol: To an ice-cold solution of Intermediate 7 (4-cyclohexanonecarboxylic acid, 5.0 g, 35 mmol) in pyridine (19 mL) and tert-butanol (27 mL) was added triclophosphorus oxychloride (POCl3, 4.7 mL, 50.6 mmol) dropwise. The reaction mixture was slowly warmed to room temperature and stirred continuously for 4 hours. Upon completion of the reaction, the crude reaction mixture was carefully poured into ice water and extracted with ethyl acetate (EtOAc). The organic layers were combined, washed with saturated brine, dried over anhydrous sodium sulfate (Na2SO4), filtered and concentrated under reduced pressure to afford Intermediate 8 (tert-butyl 4-oxocyclohexanecarboxylate, 4.0 g, 58% yield), which could be used in the next reaction without further purification. The product was confirmed by 1H NMR (400 MHz, CDCl3): δ 2.66 (tt, J = 9.6, 3.9 Hz, 1H), 2.48 (dt, J = 14.8, 5.4 Hz, 2H), 2.36 (m, 2H), 2.18 (ddd, J = 14.1, 8.7, 4.4 Hz, 2H), 2.01 (dtd, J = 14.4, 9.5, 4.8 Hz, 2H), 1.48 (s, 9H). lC/MS analysis showed a calculated value of 198.3 for m/z and a measured value of 199.1 (M + 1)+.