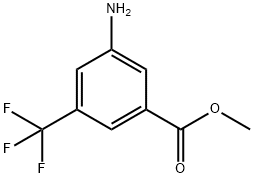
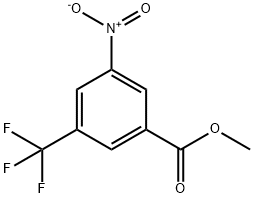
General procedure for the synthesis of methyl 3-amino-5-(trifluoromethyl)benzoate from methyl 3-nitro-5-trifluoromethylbenzoate: to a solution of methyl 3-nitro-5-(trifluoromethyl)benzoate (350 mg, 1.40 mmol) and tin(II) chloride dihydrate (1.58 g, 7.02 mmol) in methanol (20 mL) was added water (1 mL). The reaction mixture was stirred at 70 °C for 2 hours. After completion of the reaction, it was cooled to room temperature, the reaction solution was concentrated and quenched with saturated sodium bicarbonate solution. The reaction mixture was extracted with ethyl acetate (3 x 20 mL). The organic phases were combined, dried with anhydrous magnesium sulfate, filtered and concentrated to afford methyl 3-amino-5-(trifluoromethyl)benzoate (300 mg, 98% yield) as a colorless solid.1H NMR (400 MHz, CDCl3): δ 7.65 (s, 1H), 7.48 (s, 1H), 7.05 (s, 1H), 3.99 (br s, 2H), 3.91 (s, 3H).