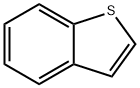
The general procedure for the synthesis of benzothiophene-2-boronic acid from trimethyl borate and benzothiophene was as follows: 144 g (554 mmol) of benzothiophene and 1,000 mL of tetrahydrofuran were added to a 2L reactor and mixed with stirring. After cooling the reaction mixture to -78 °C, 415 mL of n-butyllithium (1.6 M hexane solution) was slowly added dropwise and stirred for 1 hour at this temperature. Subsequently, the temperature of the reaction mixture was gradually brought to room temperature and kept stirring for 12 hours. After cooling again to -78 °C, 80.3 mL (664 mmol) of trimethyl borate was slowly added dropwise and stirred at room temperature for 2 hours. After completion of the reaction, the mixture was cooled to 0°C and acidified by adding 2N aqueous hydrochloric acid. Extraction was separated with ethyl acetate and distilled water and the organic layer was collected. The organic layer was concentrated under reduced pressure and filtered to give 86 g of the intermediate benzothiophene-2-boronic acid in 87.3% yield.
![Benzo[b]thien-2-ylboronic acid Structure](/CAS/GIF/98437-23-1.gif)
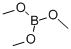

![Benzo[b]thien-2-ylboronic acid pictures](/ProductImageEN/2022-09/Small/5ce22373-f361-4809-a70a-609b2aefbd03.png)
![Benzo[b]thien-2-ylboronic acid pictures](/ProductImageEN/2023-08/Small/1861fda5-efd9-49d8-ba37-3dfd89d80ab7.jpg)