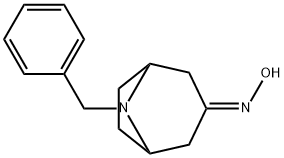
General procedure for the synthesis of exo-8-benzyl-8-azabicyclo[3.2.1]octan-3-one oxime from 8-benzyl-8-azabicyclo[3.2.1]octan-3-amine:[105] 8-benzyl-8-azabicyclo[3.2.1]octan-3-one oxime (7.65 g, 33 mmol) was dissolved in 1-pentanol (130 mL) at 120 °C. Sodium metal (7.6 g, 0.33 mol, 10.0 eq.) was added in batches over 2 hours. The mixture was stirred and heated under reflux conditions for 5 hours, then cooled to 5°C. The reaction mixture was slowly acidified with 6 M HCl until the pH reached acidic, and then the aqueous phase was further extracted with 6 M HCl (3 x 100 mL). The aqueous layer was gradually alkalized to pH 10 by addition of 5 M NaOH. the resulting aqueous solution was extracted with EtOAc (3 × 100 mL). The organic extracts were combined, dried with MgSO4, filtered and concentrated in vacuum. The crude product was purified by column chromatography (5-10% MeOH/CH2Cl2) to afford pure exo-8-benzyl-8-azabicyclo[3.2.1]octan-3-amine as a solid (2.60 g, 36%).
[1] Journal of Medicinal Chemistry, 2018, vol. 61, # 11, p. 5020 - 5033
[2] Journal of Heterocyclic Chemistry, 1982, vol. 19, # 3, p. 485 - 488
[3] Patent: WO2008/63600, 2008, A2. Location in patent: Page/Page column 11-12; 26
[4] ACS Medicinal Chemistry Letters, 2014, vol. 5, # 2, p. 133 - 137
[5] Medicinal Chemistry Research, 2018, vol. 27, # 8, p. 1906 - 1928
![3-AMINO-8-BENZYL-8-AZABICYCLO[3.2.1]OCTANE (3-EXO)- Structure](/CAS/GIF/76272-36-1.gif)
![8-Benzyl-8-azabicyclo[3.2.1]octane-3-exo-amino pictures](https://img.chemicalbook.com/ProductImageEN/2019-12/Small/189cceed-8c11-474d-8557-f38b5f694ba7.jpg)
![3-AMINO-8-BENZYL-8-AZABICYCLO[3.2.1]OCTANE (3-EXO)- pictures](/ProductImageEN/2023-10/Small/d1ec83cd-6b7f-4dd6-bfc3-180b99b9a828.jpg)
![Benzyl-8-azabicyclo[3.2.1]octan-3-exo-amine pictures](https://img.chemicalbook.com/ProductImageEN/2019-12/Small/276ef04c-3d8f-4dfd-8fc6-8072ef3bb77a.jpg)