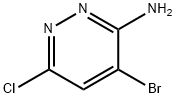
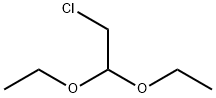
3-Amino-4-bromo-6-chloropyridazine (15.7 g, 75.3 mmol) and chloroacetaldehyde diethyl acetal (13.9 g, 90.3 mmol) were reacted in isopropanol (150 mL) in the presence of p-toluenesulfonic acid (PTSA, 17.2 g, 90.3 mmol). The reaction mixture was heated to 80 °C and maintained at this temperature for 20 hours. Upon completion of the reaction, the mixture was cooled to room temperature and subsequently concentrated under reduced pressure to remove the solvent. The concentrated residue was neutralized with saturated sodium bicarbonate solution (300 mL) and then extracted with dichloromethane (200 mL × 3). The organic layers were combined, dried with anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The residue was purified by silica gel column chromatography (200-300 mesh, petroleum ether:ethyl acetate = 3:1) to afford the target product, 8-bromo-6-chloroimidazo[1,2-b]pyridazine (17.2 g, 98% yield), as an orange solid.LC-MS analysis showed [M + H]+ m/z 231.9, 233.9, and retention time tR = 1.46 min.
![8-BROMO-6-CHLOROIMIDAZO[1,2-B]PYRIDAZINE Structure](/CAS/GIF/933190-51-3.gif)
![8-Bromo-6-chloroimidazo[1,2-b]pyridazine pictures](https://img.chemicalbook.com/ProductImageEN/2020-10/Small/bd347b42-5bdc-45ff-973e-866e7a3f8697.png)
![8-Bromo-6-chloro-imidazo[1,2-b]pyridazine pictures](https://img.chemicalbook.com/ProductImageEN/2020-4/Small/e6918841-2e4d-437f-b4de-9bf6c8a23f50.jpg)
![8-BROMO-6-CHLOROIMIDAZO[1,2-B]PYRIDAZINE pictures](https://img.chemicalbook.com/ProductImageEN/2020-1/Small/0fcef65e-a605-45d3-9af1-9c6da382b4cc.jpg)