1. Preparation of iso-PrMgCl-LiCl complex: LiCl (4.56 g, 107.6 mmol) was placed under high vacuum and dried using a hot air gun for 10 min. Under nitrogen atmosphere, iso-PrMgCl (53.8 mL, 107.6 mmol, 2M THF solution) was added to the dried LiCl, and the reaction mixture was stirred at 23 °C for 3 days to obtain a THF solution of iso-PrMgCl-LiCl complex.
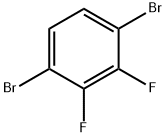
2. Synthesis of 4-bromo-2,3-difluorobenzaldehyde: A THF (5 mL) solution of 1,4-dibromo-2,3-difluorobenzene (1.29 mL, 10 mmol) was slowly added to a solution of the iso-PrMgCl-LiCl complex (5.5 mL, 11 mmol, 2.0 M THF solution) at -40 °C, making sure that the reaction temperature was maintained at below -30 °C . Subsequently, the reaction mixture was stirred at -35 to -30 °C for 1 h. The reaction was then warmed up to -7 °C to continue the reaction for 1 h. The reaction mixture was then cooled to -7 °C to continue the reaction.
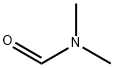
3. The reaction mixture was cooled to -30 °C and N,N-dimethylformamide (DMF, 1.00 mL, 13 mmol) was added in a single addition, at which point the temperature rose to -23 °C. The reaction mixture was stirred in the range of -25 to 15 °C for 3.5 hours.
4. Post-treatment: the reaction mixture was poured into a mixture of 1 M H2SO4 and ice, saturated with solid NaCl and extracted twice with methyl tert-butyl ether (MTBE). The organic phases were combined, dried with MgSO4, filtered and concentrated in vacuum to give 4-bromo-2,3-difluorobenzaldehyde (D-1d) 2.17 g in 98% yield as a white solid.