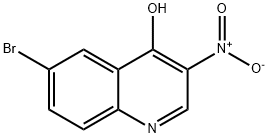
5-Bromo-2-(2-nitrovinylamino)benzoic acid (25 g, 87 mmol) was used as starting material, which was mixed with acetic anhydride (10.5 mL, 104 mmol) in acetic acid anhydride (112 mL, 1185 mmol), and the reaction was stirred for 3 hr at 120° C. The reaction was completed by filtration. After completion of the reaction, the precipitate was collected by filtration and washed with acetic acid until the filtrate was colorless. The precipitate was subsequently washed with water and dried to give the target product 6-bromo-4-hydroxy-3-nitroquinoline. Yield: 15 g (64%). The structure of the product was confirmed by 1H NMR (CDCl3, 500MHz): δ 9.275 (s, 1H), 8.611-8.615 (d, 1H, J = 2Hz), 8.100-8.118 (d, 1H, J = 9Hz), 8.026-8.048 (dd, 1H, J = 8.5Hz, 2Hz).
![Benzoic acid, 5-broMo-2-[[(1E)-2-nitroethenyl]aMino]-](/CAS/20150408/GIF/1201643-75-5.gif)


