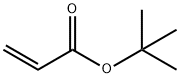
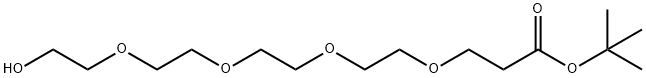
GENERAL STEPS: A piece of sodium (~1/4 cm) was added to an anhydrous tetrahydrofuran (THF, 125 mL) solution of tetraethylene glycol triacetonide (40.61 mL; 45.64 g; 235 mmol). After the sodium was fully reacted, tert-butyl acrylate (11.98 mL; 10.57 g; 82.5 mmol) was added dropwise over 20 min and the reaction mixture was stirred overnight at room temperature. Upon completion of the reaction, the pH was adjusted to 7-8 with 1N NaOH solution and the solvent was subsequently removed under reduced pressure. The residue was dissolved in a mixed solution of saturated NaHCO3 and NaCl (75 mL) and extracted with ethyl acetate (EtOAc, 3 x 100 mL). The organic layers were combined, dried with anhydrous magnesium sulfate (MgSO4), filtered, and the solvent evaporated under reduced pressure to afford tert-butyl-15-hydroxy-4,7,10,13-tetraoxapentadecanoate (21.87 g; 67.8 mmol; 82% yield, based on calculation of tert-butyl acrylate) as a colorless oil. The product was characterized by 1H-NMR (500 MHz, CDCl3, TMS) and 13C{1H}-NMR (126 MHz, CDCl3, TMS) with the following data: 1H-NMR δ [ppm] = 3.77-3.57 (m, 18H, OCH2); 3.01 (bs, 1H, OH); 2.51 (t, 2H, J = 6.6 Hz, CH2COO'Bu); 1.45 (s, 9H, C(CH3)3); 13C{1H}-NMR δ [ppm] = 171.1 (COO); 80.7 (C(CH3)3); 72.6 / 70.8 / 70.7 / 70.6 / 70.5 / 67.0 (OCH2); 61.9 (HOCH2); 36.4 ( CH2CO); 28.2 (C(CH3)3).
![Bis[2-(2-hydroxyethoxy)ethyl] ether](/CAS/GIF/112-60-7.gif)