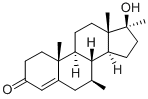
As described in US Patent 3,029,263, one possibility is a multistep synthesis
starting from 3β,17β-dihydroxy-17α-methyl-5-androstene.
Alternatively, as described in US Patent 3,341,557, 6-dehydro-17-
methyltestosterone may be used as the starting material. A mixture of 0.4 g
of cuprous chloride, 20 ml of 4M methylmagnesium bromide in ether and 60
ml of redistilled tetrahydrofuran was stirred and cooled in an ice bath during
the addition of a mixture of 2.0 g of 6-dehydro-17-methyltestosterone, 60 ml
of redistilled tetrahydrofuran and 0.2 g of cuprous chloride. The ice bath was
removed and stirring was continued for four hours. Ice and water were then
carefully added, the solution acidified with 3 N hydrochloric acid and extracted
several times with ether. The combined ether extracts were washed with a
brine-sodium carbonate solution, brine and then dried over anhydrous
magnesium sulfate, filtered and then poured over a 75-g column of
magnesium silicate (Florisil) packed wet with hexanes (Skellysolve B). The
column was eluted with 250 ml of hexanes, 0.5 liter of 2% acetone, two liters
of 4% acetone and 3.5 liters of 6% acetone in hexanes.
Four 250-ml fractions were collected followed by 150 ml fractions. The
residues from fractions 8 to 16 were combined and rechromatographed over a
125-g column of magnesium silicate. The solumn was eluted with 6% acetone
in hexanes which was collected in 150 ml portions. Fractions 18 to 29 were
combined and dissolved in acetone, decolorized with charcoal, and
recrystallized from acetone. One gram of a crystalline mixture of the 7epimers
of 7,17-dimethyltestosterone was obtained melting at 120° to 140°C.