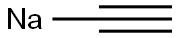
SODIUM ACETYLIDE
- Product NameSODIUM ACETYLIDE
- CAS1066-26-8
- MFC2HNa
- MW48.02
- EINECS213-908-9
- MOL File1066-26-8.mol
Chemical Properties
| Boiling point | -22.2°C |
| Density | 0,865 g/cm3 |
| Flash point | 85 °F |
| storage temp. | Refrigerator (+4°C) + Flammables area |
| form | Low Melting Solid or Crystalline Powder |
| color | White to pale yellow |
| Water Solubility | Reacts with water. |
| Sensitive | Air & Moisture Sensitive |
| BRN | 3587232 |
| InChI | 1S/C2H.Na/c1-2;/h1H; |
| InChIKey | SLBNQJAGYKQCKI-UHFFFAOYSA-N |
| SMILES | [Na]C#C |
| CAS DataBase Reference | 1066-26-8(CAS DataBase Reference) |
| EPA Substance Registry System | Sodium acetylide (1066-26-8) |
Safety Information
| Hazard Codes | F,Xn,C |
| Risk Statements | 10-15-20/21-38-34-14/15 |
| Safety Statements | 36/37-43-45-36/37/39-26 |
| RIDADR | UN 3399 4.3/PG 1 |
| WGK Germany | 3 |
| TSCA | TSCA listed |
| HazardClass | 4.3 |
| PackingGroup | II |
| HS Code | 29310099 |
| Storage Class | 4.3 - Hazardous materials which set free flammable gases upon contact with water |
| Hazard Classifications | Acute Tox. 4 Dermal Acute Tox. 4 Inhalation Aquatic Chronic 3 Asp. Tox. 1 Eye Dam. 1 Flam. Liq. 3 Skin Corr. 1B STOT RE 2 Inhalation STOT SE 3 Water-react 2 |
