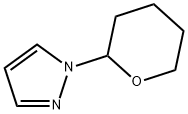
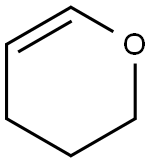
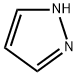
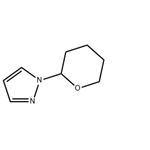
General procedure for the synthesis of 1-(tetrahydro-2H-pyran-2-yl)-1H-pyrazole from 3,4-dihydro-2H-pyran and pyrazole: cf. Example 1 [1-(tetrahydro-2H-pyran-2-yl)-1H-pyrazol-5-yl]boronic acid. Pyrazole (14.3 g, 0.21 mol) was mixed with 3,4-dihydro-2H-pyran (29 mL, 0.32 mol) and trifluoroacetic acid (0.1 mL, 0.0013 mol) and heated under reflux conditions for 5 hours. Upon completion of the reaction, the mixture was cooled to room temperature, followed by the addition of sodium hydride (60% dispersed in mineral oil, 0.2 g, 0.008 mol) and stirred for 10 min. Finally, the reaction mixture was purified by reduced pressure distillation (60-65 °C, 0.5-1 mmHg) to afford the target product 1-(tetrahydropyran-2-yl)-1H-pyrazole (30.8 g, 96% yield).
[1] Tetrahedron Letters, 2006, vol. 47, # 27, p. 4665 - 4669
[2] Tetrahedron Letters, 2007, vol. 48, # 23, p. 4123 - 4126
[3] Journal of Organic Chemistry, 2016, vol. 81, # 4, p. 1718 - 1722
[4] Patent: WO2017/66606, 2017, A1. Location in patent: Page/Page column 49
[5] Patent: WO2007/138072, 2007, A2. Location in patent: Page/Page column 54