Fluorescent brightener
1929 P. Krais found that aesculetin can emit fluoresces under ultraviolet light with the fabric, after being immersed in an aqueous solution, obtaining whitening effect. The first commercially available fluorescent brightener was the derivatives of the 4, 4'-bis-triazine stilbene-2, 2'-disulfonic acid developed by the Federal Republic of Germany Bayer company in 1940. Since then, people have further developed a number of fluorescent brightener agents, and thus becoming self-contained series dyes.
Fluorescent brightener is commonly known as "white dye", also known as optical brighteners, briefly called brighteners. It is a colorless organic compound that can stimulate fluorescence under ultraviolet light. Its brightening effect is through the principle of the optically complementary color, making yellowing substances, after subjecting to fluorescent brightener treatment, not only can reflect visible light, but also absorb UV light outside the visible light into visible light of blue/yellow or cyan to be reflected. It is organic matters that can improve the whiteness of the fabrics and paper. Brighteners can absorb the ultraviolet in 330 to 360 nm wavelength with the reflected light being at a blue/violet light of wavelength of 400 to 440 nm, coinciding with the yellow light reflected from the fabric undergoing bleaching but still exhibits yellow color as mutually complementary colors. Therefore, these two colors add with each other and become white so that the fabric becomes white. After the whitening process, due to that there is a portion of ultraviolet light becoming visible light to be reflected out with the total reflection amount increased compared to non-processing fabric, the brightness of the treated fabric also increases, becoming whiter. It can also increase the vividness of the colored fabrics. Fluorescent whitening, being different with chemical bleaching, would not destroy the fabric color and intensity. However, for fabric and paper of darker background, if they undergo direct brightening treatment without bleaching, can’t obtain good results.
The organic compound taken as fluorescent brightening agents should having at least four conjugated double bonds, such as -C = C-C = C -C = C-C = C- or -N = C-C = C- C = N -C = C- in its molecule structure. The common structure of chromophore contains four kinds:
① Stilbene type: such as brighteners VBL. It is mainly used for the brightening of cotton fibers and being taken as the brightening component in advanced synthetic detergent.
② Coumarin type, such as brighteners WS. It has a strong blue fluorescence, being the brighter of polyamide, wool and silk.
③ Azole type; It is mainly for the brightening of wool, polyamide and acrylon.
④ Naphthalimide type: such as APL. It can be applied to the brightening of the polyamide, polyester and PVC.
Brightener is mainly used in textile industry to improve the whiteness of a variety of natural and synthetic fabrics and enhance shine. In recent years, its application in the fields of paper, plastics, leather, soap, synthetic detergent and other daily necessities in is also growing rapidly.
- Structure:
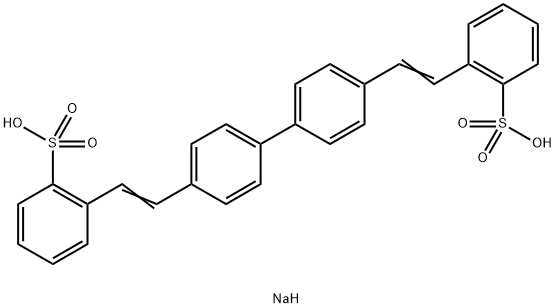
- Chemical Name:Disodium 4,4'-bis(2-sulfostyryl)biphenyl
- CAS:27344-41-8
- MF:C28H23NaO6S2
- Structure:
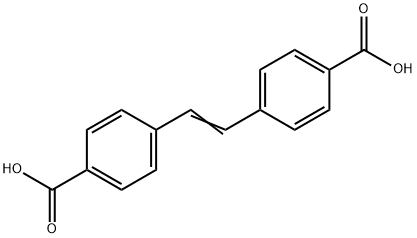
- Chemical Name:4,4'-Stilbenedicarboxylic acid
- CAS:100-31-2
- MF:C16H12O4
- Structure:
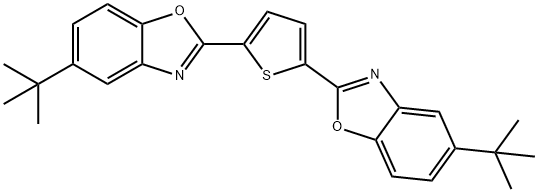
- Chemical Name:2,5-Bis(5-tert-butyl-2-benzoxazolyl)thiophene
- CAS:7128-64-5
- MF:C26H26N2O2S
- Structure:
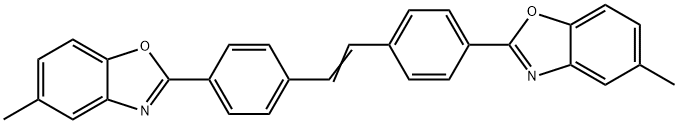
- Chemical Name:4,4'-Bis(5-methyl-2-benzoxazolyl)stilbene
- CAS:2397-00-4
- MF:C30H22N2O2
- Structure:
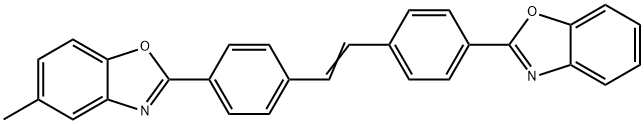
- Chemical Name:Fluorescent Brightener KS-N
- CAS:5242-49-9
- MF:C29H20N2O2
- Structure:
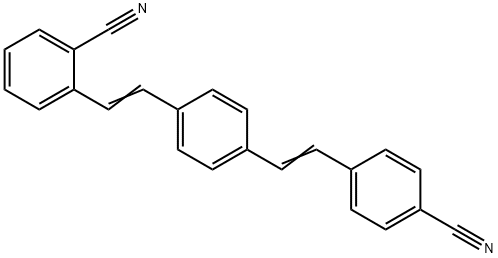
- Chemical Name:1-(2-Cyanostyryl)-4-(4-cyanostyryl)benzene
- CAS:13001-38-2
- MF:C24H16N2
- Structure:
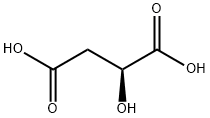
- Chemical Name:L-Malic acid
- CAS:97-67-6
- MF:C4H6O5
- Structure:
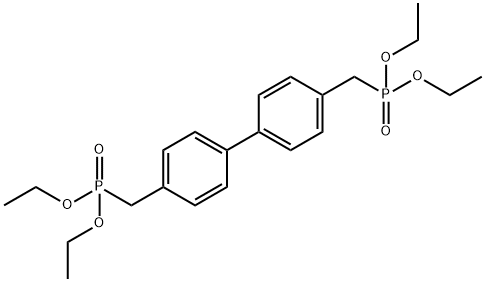
- Chemical Name:4,4-Bis(diethylphosphonomethyl)biphenyl
- CAS:17919-34-5
- MF:C22H32O6P2
- Structure:
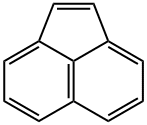
- Chemical Name:Acenaphthylene
- CAS:208-96-8
- MF:C12H8
- Structure:
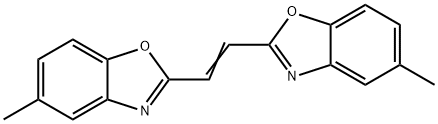
- Chemical Name:Fluorescent Brightener 135
- CAS:1041-00-5
- MF:C18H14N2O2
- Structure:
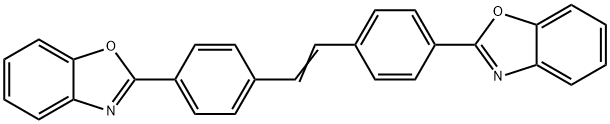
- Chemical Name:Optical brightener OB-1
- CAS:1533-45-5
- MF:C28H18N2O2
- Structure:
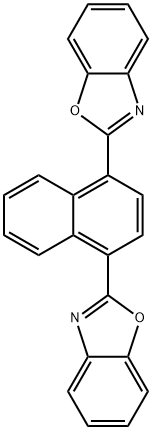
- Chemical Name:Fluorescent Brightener 367
- CAS:5089-22-5
- MF:C24H14N2O2
- Structure:
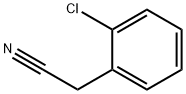
- Chemical Name:2-(2-Chlorophenyl)acetonitrile
- CAS:2856-63-5
- MF:C8H6ClN
- Structure:
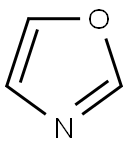
- Chemical Name:Oxazole
- CAS:288-42-6
- MF:C3H3NO
- Structure:
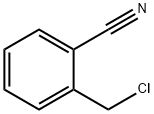
- Chemical Name:2-(Chloromethyl)benzonitrile
- CAS:612-13-5
- MF:C8H6ClN
- Structure:
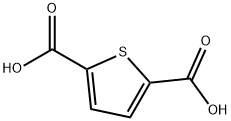
- Chemical Name:2,5-Thiophenedicarboxylic acid
- CAS:4282-31-9
- MF:C6H4O4S
- Structure:
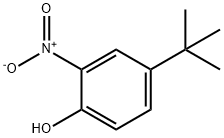
- Chemical Name:4-tert-Butyl-2-nitrophenol
- CAS:3279-07-0
- MF:C10H13NO3
- Structure:
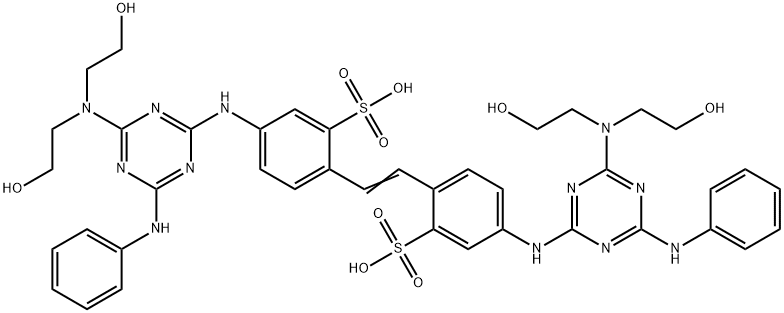
- Chemical Name:FLUORESCENT BRIGHTENER 28
- CAS:4404-43-7
- MF:C40H44N12O10S2
- Structure:
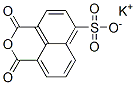
- Chemical Name:4-Sulfo-1,8-naphthalic anhydride potassium salt
- CAS:71501-16-1
- MF:C12H5KO6S
- Structure:
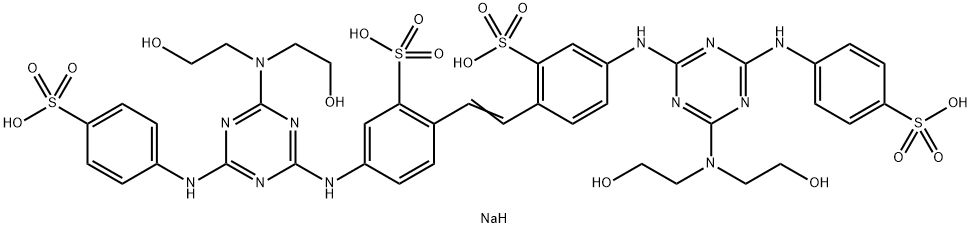
- Chemical Name:Fluorescent Brightener 220
- CAS:16470-24-9
- MF:C40H45N12NaO16S4
- Structure:
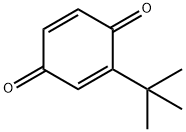
- Chemical Name:2-tert-Butyl-1,4-benzoquinone
- CAS:3602-55-9
- MF:C10H12O2
- Structure:
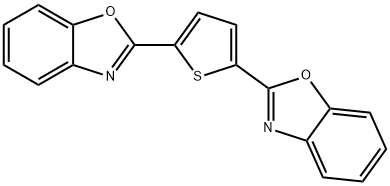
- Chemical Name:Fluorescent Brightener 185
- CAS:2866-43-5
- MF:C18H10N2O2S
- Structure:
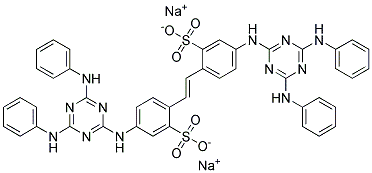
- Chemical Name:Fluorescent Brightener 9
- CAS:133-66-4
- MF:C44H34N12Na2O6S2
- Chemical Name:Fluorescent Whitening Agent VBL
- CAS:
- MF:
- Chemical Name:Fluorescent Brightener BC
- CAS:
- MF:C32H26N12Na2O6S2
- Chemical Name:Fluorescent Brightener BBU
- CAS:
- MF:
- Structure:
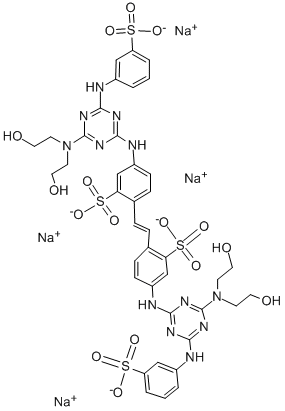
- Chemical Name:Fluorescent Brightener 87
- CAS:12768-91-1
- MF:C40H40N12O16S4.4Na
- Structure:
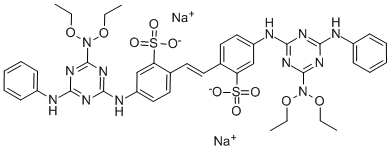
- Chemical Name:Fluorescent Brightener 113
- CAS:12768-92-2
- MF:C40H42N12O10S2.2Na
- Chemical Name:Fluorescent Brightener Pf
- CAS:
- MF:C18H14O2N2
- Chemical Name:Fluorescent Brightener 33
- CAS:61902-19-0
- MF:
- Structure:
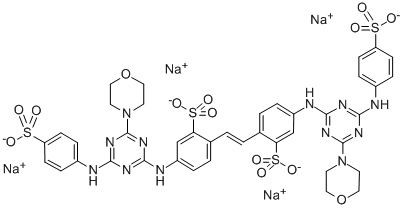
- Chemical Name:Fluorescent Brightener 210
- CAS:28950-61-0
- MF:C40H36N12Na4O14S4
- Chemical Name:Fluorescent Whitener ER
- CAS:
- MF:
- Structure:
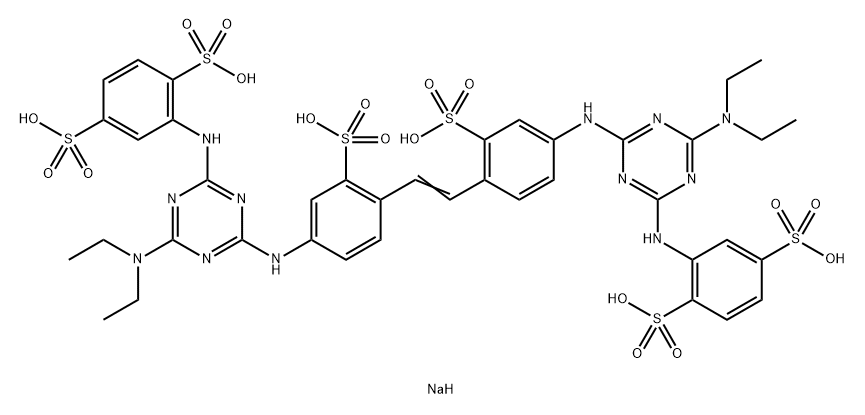
- Chemical Name:Fluorescent Brightener 357
- CAS:41098-56-0
- MF:C40H45N12NaO18S6
- Structure:
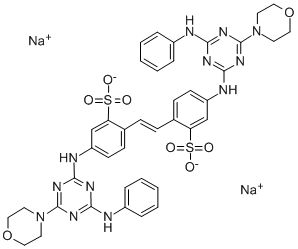
- Chemical Name:Fluorescent brightener 71
- CAS:16090-02-1
- MF:C40H38N12Na2O8S2
- Structure:
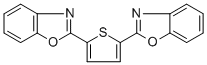
- Chemical Name:Fluorescent brightener 185
- CAS:12224-41-8
- MF:C18H10N2O2S
- Structure:
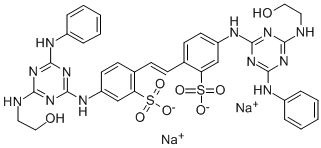
- Chemical Name:Fluorescent brightener 85
- CAS:12224-06-5
- MF:C36H34N12Na2O8S2
- Structure:
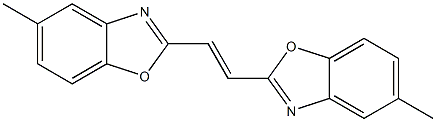
- Chemical Name:Fluorescent brightener 135
- CAS:12224-12-3
- MF:C18H14N2O2
- Structure:
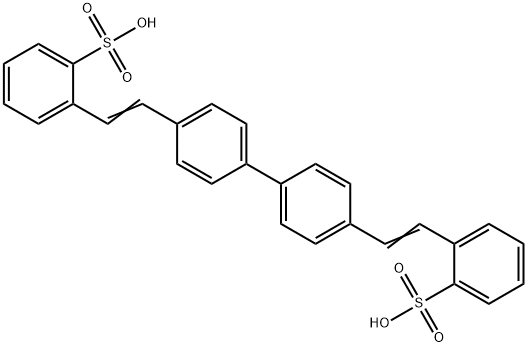
- Chemical Name:C.I. 482200
- CAS:38775-22-3
- MF:C28H22O6S2
- Structure:
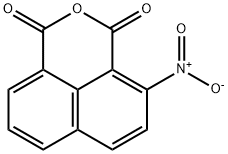
- Chemical Name:4-Nitronaphthalene-1,8-dicarboxylic anhydride
- CAS:34087-02-0
- MF:C12H5NO5
- Chemical Name:Fluorescent Brightener 191
- CAS:12270-53-0
- MF:C30H20N6Na2O6S2
- Chemical Name:Fluorescent Brightener 204
- CAS:76482-77-4
- MF:C32H26N12Na2O6S2
- Structure:
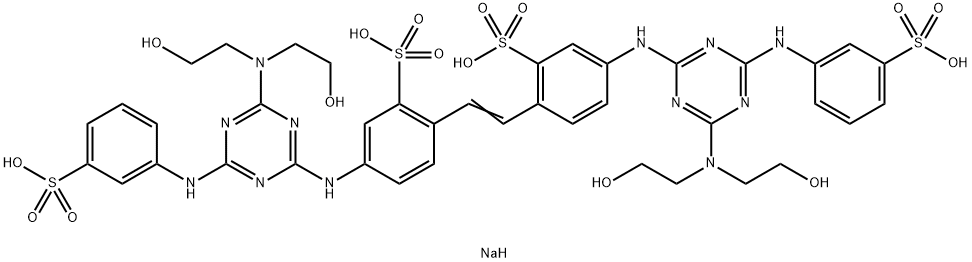
- Chemical Name:Fluorescent brightener 24
- CAS:12224-02-1
- MF:C40H45N12NaO16S4
- Chemical Name:Fluorescent brightener PF-3
- CAS:
- MF:C18H14N2O2
- Structure:
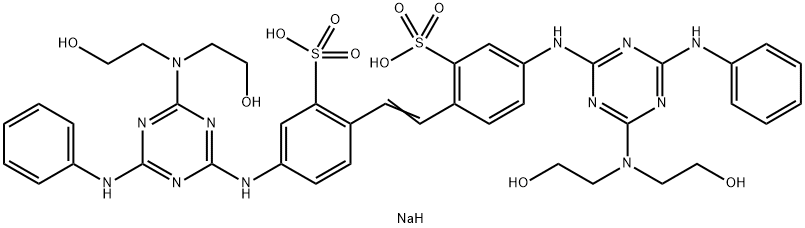
- Chemical Name:Fluorescent Brightener 28
- CAS:4193-55-9
- MF:C40H45N12NaO10S2
- Structure:
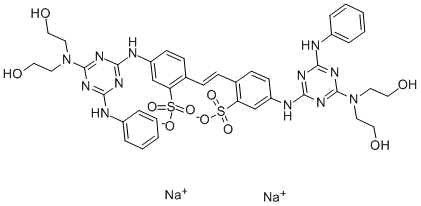
- Chemical Name:Fluorescent brightener 86
- CAS:12224-07-6
- MF:C40H42N12O10S2.2Na
- Structure:
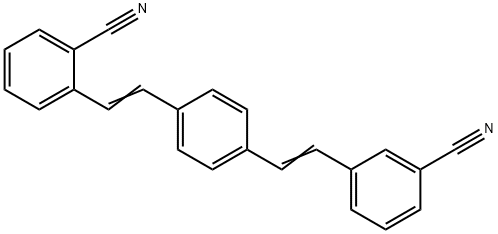
- Chemical Name:Fluorescent Brightener ER-III
- CAS:79026-03-2
- MF:C24H16N2
- Structure:
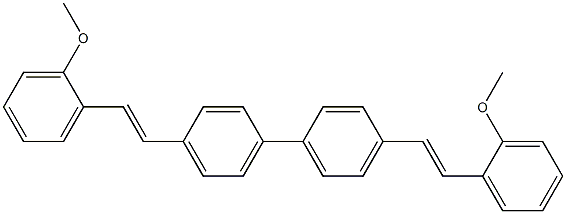
- Chemical Name:Fluorescent brightener 378
- CAS:164908-53-6
- MF:C30H26O2
- Chemical Name:Fluorescent brightener BA
- CAS:
- MF:
- Structure:
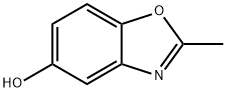
- Chemical Name:5-BENZOXAZOLOL, 2-METHYL-
- CAS:23997-94-6
- MF:C8H7NO2
- Structure:
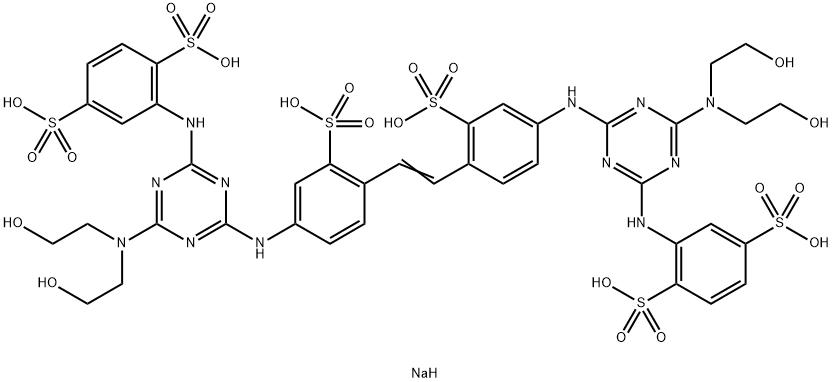
- Chemical Name:Fluorescent Brightener 264
- CAS:68971-49-3
- MF:C40H45N12NaO22S6
- Chemical Name:Fluorescent Brightener 353
- CAS:55585-28-9
- MF:
- Structure:
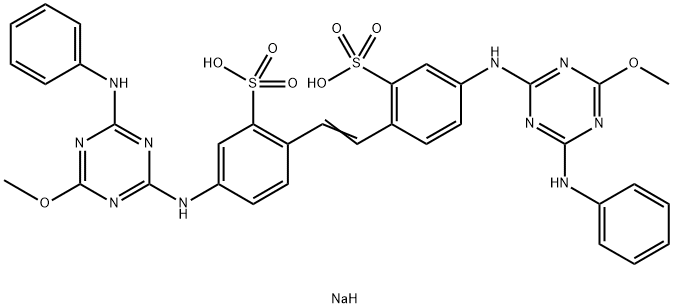
- Chemical Name:Disodium 4,4'-bis[(4-anilino-6-methoxy-1,3,5-triazin-2-yl)amino]stilbene-2,2'-disulphonate
- CAS:3426-43-5
- MF:C34H31N10NaO8S2
- Structure:
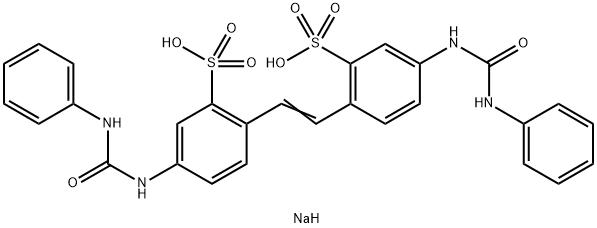
- Chemical Name:Fluorescent Brightener 30
- CAS:2606-93-1
- MF:C28H25N4NaO8S2
- Chemical Name:BRIGHTENERS
- CAS:
- MF:
- Chemical Name:Fluorescent Brightener
- CAS:
- MF:
- Chemical Name:Fluorescent brightener CBS
- CAS:
- MF:C28H22O6S2
- Chemical Name:fluorescent whitening agent PRS
- CAS:
- MF:C34H28N10Na2O8S2
- Chemical Name:Fluorescent brightener EB
- CAS:
- MF:
- Structure:
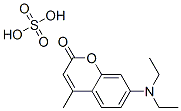
- Chemical Name:MDAC)
- CAS:12224-03-2
- MF:C14H17NO2
- Chemical Name:Fluorescent brightener PRA
- CAS:
- MF:
- Chemical Name:Fluorescent brightener NT-1
- CAS:
- MF:
- Chemical Name:Fluorescent brightener A
- CAS:
- MF:
- Chemical Name:Fluorescent brightener PSD
- CAS:
- MF:
- Structure:
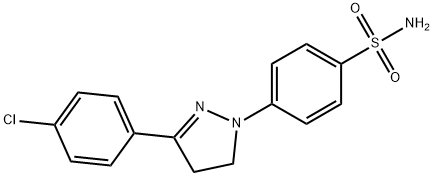
- Chemical Name:Fluorescent Brightener 121
- CAS:2744-49-2
- MF:C15H14ClN3O2S
- Chemical Name:Fluorescent Brightener 119
- CAS:12270-52-9
- MF:
- Chemical Name:Fluorescent Brightener 54
- CAS:12768-89-7
- MF:C21H16ClN2NaO3S
- Structure:
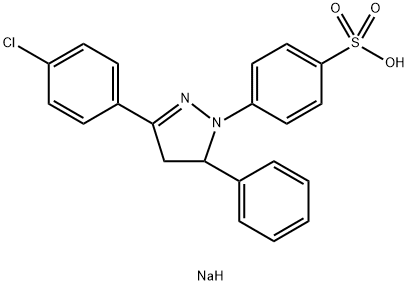
- Chemical Name:sodium 4-[3-(4-chlorophenyl)-4,5-dihydro-5-phenyl-1H-pyrazol-1-yl]benzenesulphonate
- CAS:40691-09-6
- MF:C21H18ClN2NaO3S
- Structure:
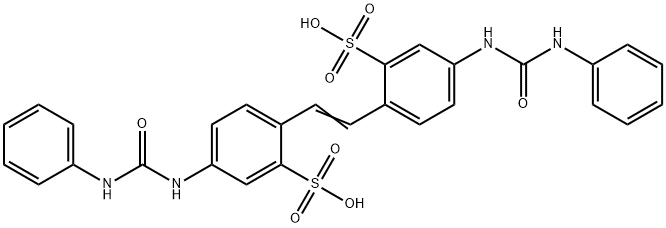
- Chemical Name:4,4'-bis(3-phenylureido)-2,2'-stilbenedisulphonic acid
- CAS:25738-35-6
- MF:C28H24N4O8S2
- Chemical Name:Fluorescent Whitening Agent EBF
- CAS:
- MF:
- Chemical Name:Fluorescent Brightener XD
- CAS:
- MF:
- Chemical Name:Fluorescent brightener PSM
- CAS:
- MF:
- Chemical Name:Fluorescent brightener APL
- CAS:
- MF:
- Chemical Name:Fluorescent brightener PB
- CAS:
- MF:C18H10N2O2S
- Chemical Name:Fluorescent Brightener 363
- CAS:95078-19-6
- MF:
- Structure:
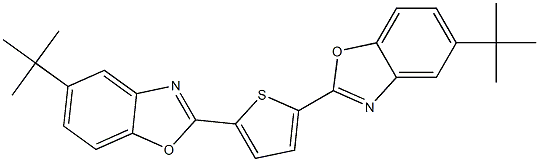
- Chemical Name:Fluorescent Brightener 184
- CAS:12224-40-7
- MF:C26H26N2O2S
- Chemical Name:Fluorescent Brightener VBU
- CAS:
- MF:C40H42N12Na4O12S4
- Chemical Name:Fluorescent brightener PMB
- CAS:
- MF:
- Structure:
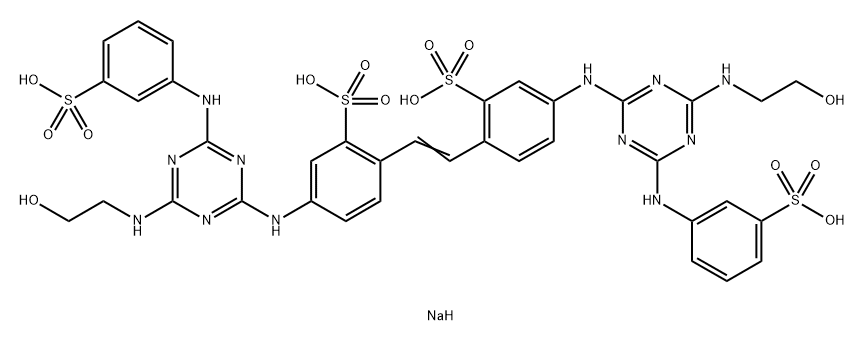
- Chemical Name:Fluorescent Brightener 251
- CAS:16324-27-9
- MF:C36H37N12NaO14S4
- Chemical Name:Fluorescent Brightener 263
- CAS:99549-42-5
- MF:C40H38N12Na6O18S6
- Structure:
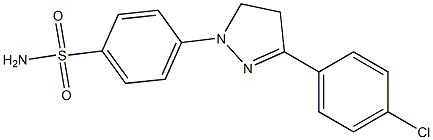
- Chemical Name:Fluorescent brightener 121
- CAS:12224-11-2
- MF:C15H14ClN3O2S
- Chemical Name:fluorescent whitening agent SWN
- CAS:
- MF:C14H17NO2
- Chemical Name:Fluorescent brightener RKH
- CAS:
- MF:
- Chemical Name:Fluorescent brightener OB-C
- CAS:
- MF:
- Chemical Name:Fluorescent Brightener DCB
- CAS:
- MF:C15H14ClN2O2S
- Chemical Name:Fluorescent Brightener VBA
- CAS:
- MF:C40H36N12O14S4Na4
- Chemical Name:Fluorescent whitening agent for plastic
- CAS:
- MF:
- Chemical Name:Fluorescent brightener KB
- CAS:
- MF:
- Chemical Name:Fluorescent brightener V4BM
- CAS:
- MF:
- Chemical Name:Fluorescent brightener PSW-201
- CAS:
- MF:
- Chemical Name:Fluorescent brightener KOB
- CAS:
- MF:
- Chemical Name:Fluorescent Brightener 1
- CAS:15339-39-6
- MF:Unspecified
- Chemical Name:Fluorescent Brightener BSL
- CAS:
- MF:
- Chemical Name:Fluorescent Brightener BF
- CAS:
- MF:C1634N12Na2O8S2
- Chemical Name:Fluorescent brightener HDT-1
- CAS:
- MF:
- Chemical Name:Fluorescent brightener HL
- CAS:
- MF:
- Chemical Name:Fluorescent brightener PS-1
- CAS:
- MF:
- Chemical Name:Fluorescent brightener BSU
- CAS:
- MF:
- Chemical Name:Fluorescent brightener CEF
- CAS:
- MF:
- Chemical Name:Fluorescent brightener OB-P
- CAS:
- MF:
- Structure:
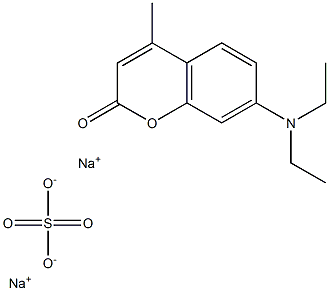
- Chemical Name:CALCOFLUOR WHITE RWP
- CAS:8066-05-5
- MF:C14H17NNa2O6S