Synthesis
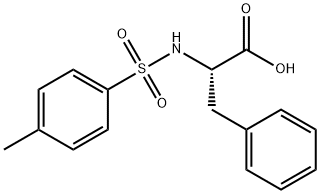
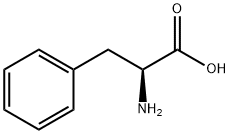
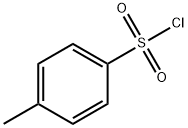
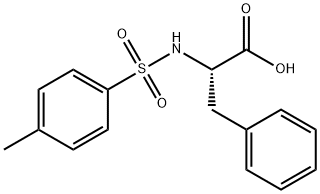
GENERAL STEPS: Sodium carbonate (Na2CO3, 1.590 g, 15 mmol) was added dropwise to an aqueous solution (15 mL) of L-phenylalanine (12.5 mmol) under continuous stirring conditions until complete dissolution. Subsequently, the reaction system was cooled to -5°C and p-toluenesulfonyl chloride (15 mmol) was slowly added in four portions over 1 hour. The reaction mixture was continued to be stirred at room temperature for 4 h, during which the progress of the reaction was monitored by thin layer chromatography (TLC, unfolding agent was methanol/dichloromethane, 1:9). Upon completion of the reaction, the mixture was acidified with 20% aqueous hydrochloric acid to pH 2. The precipitated solid was collected by diafiltration and washed with pH 2.2 buffer. Finally, the purified product (S)-2-(4-methylphenylsulfonamido)-3-phenylpropionic acid was placed in a desiccator and dried using fused silica gel as desiccant.
References
[1] European Journal of Medicinal Chemistry, 2017, vol. 135, p. 349 - 369
[2] PLoS ONE, 2018, vol. 13, # 1,
[3] RSC Advances, 2015, vol. 5, # 24, p. 18751 - 18760
[4] European Journal of Organic Chemistry, 2005, # 5, p. 934 - 938
[5] Journal of Organic Chemistry, 2012, vol. 77, # 9, p. 4375 - 4384