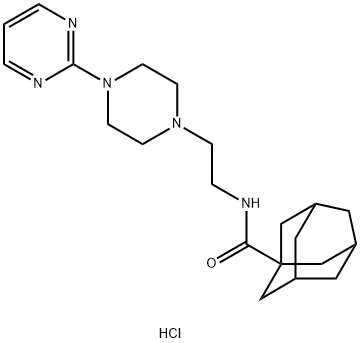
Manufacturing Process
To a stirred solution of [4-(2-pyrimidinyl)piperazino]ethylamine (2.0 g, 0.01
mol) in 50 ml of methylene chloride, adamantane-1-carboxylic acid chloride
(3.6 g, 0.018 mol) and triethylamine (2.9 g, 0.015 mol) were added. Stirring
was continued at room temperature overnight. The methylene chloride
solution was washed with water, dried over anhydrous sodium sulfate and
evaporated under reduced pressure. The remaining residue was subjected to
preparative HPLC. The residue was dissolved in ethyl acetate (10 ml) and
subjected to flash chromatography using a 9 inch column of silica gel and
ethyl acetate as the eluent. The N-[2-[4-(2-pyrimidinyl)-1-
piperazinyl]ethyl]tricyclo[3.3.1.1(3,7)]
decane-1-carboxamide was separated.
In practice it is usually used as hydrochloride.