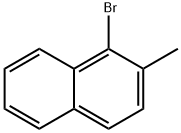
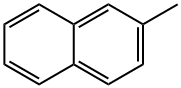
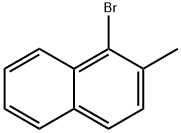
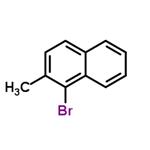
Chemical Properties
CLEAR YELLOW LIQUID
Uses
1-Bromo-2-methylnaphthalene was used as test compound in determination of halogenated hydrocarbons at trace levels by supercritical fluid chromatography-microwave-induced plasma mass spectrometry.
Uses
1-Bromo-2-methylnaphthalene is used as test compound in determination of halogenated hydrocarbons at trace levels by supercritical fluid chromatography-microwave-induced plasma mass spectrometry. Dynamic phosphorescence quenching of 1-bromo-2-methylnaphthalene without deoxygenation has been used for selective sensing of Cu(II) at ngm levels.
General Description
Dynamic phosphorescence quenching of 1-bromo-2-methylnaphthalene without deoxygenation has been used for selective sensing of Cu(II) at ngml
-1 levels. It undergoes asymmetric cross-coupling reaction with its corresponding Grignard reagent using a nickel catalyst to form non-racemic 2,2′-dimethyl-1,1′-binaphthyl.
Synthesis
General procedure for the synthesis of 1-bromo-2-methylnaphthalene from 2-methylnaphthalene:
1. To a stirred mixed solution of carbon tetrachloride (20 mL) and cyclopentene (2.94 g, 43.1 mmol) was added sequentially N-bromosuccinimide (8.06 g, 45.3 mmol) and a catalytic amount of benzoyl peroxide under argon protection. The reaction mixture was heated and stirred at reflux for 1 hour. Upon completion of the reaction, the resulting solid was removed by filtration. The filtrate was concentrated in vacuum to give 3-bromocyclopentene, which was used in the next step of the reaction without further purification.
2. To a stirred suspension of sodium hydride (60% dispersed in mineral oil, 2.60 g, 65.0 mmol) in tetrahydrofuran (10 mL) was slowly added a solution of malononitrile (5.70 g, 86.2 mmol) in tetrahydrofuran (5 mL) at 0 °C and under argon atmosphere. The suspension was stirred at room temperature for 1 hour. Subsequently, a tetrahydrofuran (5 mL) solution of 3-bromocyclopentene was slowly added at 0 °C and stirring was continued at room temperature for 2 hours. After completion of the reaction, ether and saturated saline were added, the organic layer was separated, dried over anhydrous sodium sulfate, filtered and concentrated in vacuum. The residue was purified by silica gel column chromatography (eluent: hexane-ethyl acetate) to afford 3-(dicyanomethyl)cyclopentene (0.889 g, 6.73 mmol, 16% yield).
3. To a stirred solution of carbon tetrachloride (60 mL) containing 2-methylnaphthalene (20.1 g, 141 mmol), a catalytic amount of iron powder and iodine was slowly added a solution of carbon tetrachloride (10 mL) with bromine (22.5 g, 7.2 mL, 141 mmol) at 0 °C. The reaction mixture was stirred for 3 hours. Upon completion of the reaction, the reaction was quenched by the addition of saturated aqueous sodium thiosulfate solution. The organic layer was washed with saturated saline, dried over anhydrous sodium sulfate, filtered and concentrated in vacuum. The residue was purified by silica gel column chromatography (eluent: hexane) to afford 1-bromo-2-methylnaphthalene (29.3 g, 133 mmol, 94% yield). The product was brown liquid; 1H NMR (300 MHz, CDCl3) δ 2.58 (s, 3H), 7.26-8.24 (m, 6H) ppm.
References
[1] Bulletin of the Chemical Society of Japan, 1989, vol. 62, # 2, p. 439 - 443
[2] Journal of Heterocyclic Chemistry, 2018, vol. 55, # 3, p. 670 - 684
[3] Angewandte Chemie - International Edition, 2018, vol. 57, # 39, p. 12869 - 12873
[4] Angew. Chem., 2018, vol. 130, p. 13051 - 13055,5
[5] Tetrahedron, 2009, vol. 65, # 22, p. 4429 - 4439