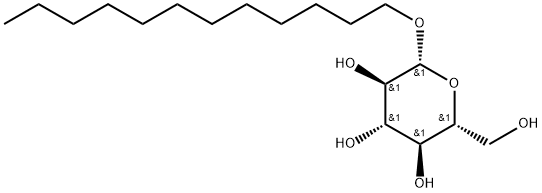
Synthesis
A synthetic method of n-dodecyl-beta-D-glucopyranoside, comprising the steps of:
1) dissolving fully acetylated glucopyranoside, n-dodecyl alcohol and anhydrous tin tetrachloride in anhydrous dichloromethane, stirring the reaction for 20 minutes at room temperature, washing it with saturated aqueous potassium carbonate, collecting the organic phase, and distilling it under reduced pressure to obtain 1-n-dodecyl-2,3,4,6-tetraacetyl--D-glucopyranoside; the molar ratio of fully acetylated glucopyranoside, n-dodecyl alcohol and anhydrous tin tetrachloride was: 1:1.2:1.2; the ratio of fully acetylated glucopyranoside to anhydrous dichloromethane was 100g:550ml;
2) Dissolve 1-n-dodecyl-2,3,4,6-tetraacetyl--D-glucopyranoside in methanol, add sodium methoxide to adjust the pH to 9, and reacted at room temperature for 1.5 h. The reaction was adjusted to neutrality with a strongly acidic cation exchange resin Dowex-50, filtered, and the filtrate was evaporated out of the solvent, dried, and detected by NMR to give n-dodecyl-beta-D-glucopyranoside. 1-n-dodecyl-2,3,4,6-tetraacetyl--D-glucopyranoside to methanol ratio was 100 g:520 ml.
The yield was 57%. And the beta configuration was >97%.