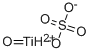Chemical Properties
White, cake-like solid. Highly acidic,
similar to 50% sulfuric acid, typical composition
20% TiO2, 50% sulfuric acid, 30% water,
hygroscopic. Soluble in water; solutions
hydrolyze readily unless protected from heat and
dilution.
Uses
TiOSO
4 (titanium (IV) oxysulfate) was used for the preparation of stable amorphous titanium oxide (TiO
2) hollow microspheres.
Uses
In tanning; in production of pigment grade and of anatase-structured titanium dioxide.
Production Methods
Pure, multiple-distilled TiCl4 is added in drops (vigorous stirring) to the stoichiometric quantity of 50% H2SO4. The precipitate formed after each addition should dissolve completely before the next portion of TiCl4 is added. After adding about 3/4 of the TiCl4, the liquid turns into a viscous, yellowish solution. It is diluted with 1/5 its volume of water, and the dropwise addition of TiCl4 is completed. The resultant solution, which is still highly concentrated in HCl, is evaporated to dryness in a water bath. The residue is pulverized, dried, and freed of HCl by heating it for several days at 80-100°C in a drying pistol at a pressure of a few mm. The sulfate obtained in this manner (TiO2: SO3 = 1:1.07) is colourless and free of HCl. It is hygroscopic and soluble in water, yielding a clear solution.
General Description
A colorless or violet colored liquid. Dilution releases heat and may cause precipitation of solid titanium sulfate. Corrosive to metals and tissue. TITANIUM OXYSULFATE is used to make pigments and in dyeing.
Air & Water Reactions
Can generate heat on dilution with water.
Reactivity Profile
A highly acidic liquid.
Hazard
Irritant to skin and tissue.
Health Hazard
TOXIC; inhalation, ingestion or skin contact with material may cause severe injury or death. Contact with molten substance may cause severe burns to skin and eyes. Avoid any skin contact. Effects of contact or inhalation may be delayed. Fire may produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution.
Fire Hazard
Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Some are oxidizers and may ignite combustibles (wood, paper, oil, clothing, etc.). Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated.
reaction suitability
reagent type: catalyst
core: titanium
Safety Profile
A corrosive irritant to
skin, eyes, and mucous membranes. When
heated to decomposition it emits toxic
fumes of SOx. See also TITANIUM
COMPOUNDS and SULFATES.
Purification Methods
Dissolve it in water, filter and crystallise it three times from boiling 45% H2SO4, washing with EtOH to remove excess acid, then with Et2O. Dry it in air for several hours, then in an oven at 105-110o. [Hixson & Fredrickson Ind Eng Chem 37 678 1945.]
Structure and conformation
The structure of titanium(IV) oxysulfate consists of dense polymeric network with tetrahedral sulfur and octahedral titanium centers. The six ligands attached to titanium are derived from four different sulfate moieties and a bridging oxide.