Synthesis
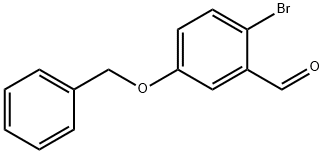
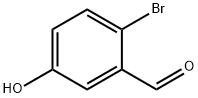
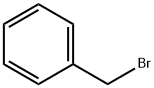
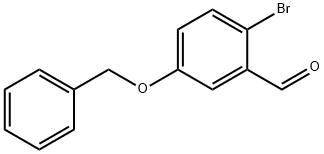
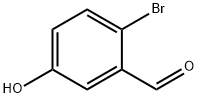
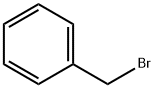
Example 396: Synthesis of (1E,6E)-1-(5-benzyloxy-2-bromophenyl)-7-(4-hydroxyphenyl)hepta-1,6-diene-3,5-dione (CU531); (1) Synthesis of 5-benzyloxy-2-bromobenzaldehyde; To 2-bromo-5-hydroxybenzaldehyde (100 mg, 0.50 mmol), potassium carbonate (138 mg, 1.00 mmol) and tetrabutylammonium iodide (18 mg, 0.05 mmol) in a suspension of 1.0 mL of dry N,N-dimethylformamide, benzyl bromide (89 μL, 0.74 mmol) was slowly added at 0 °C. The reaction mixture was stirred overnight at room temperature, then diluted with ether, washed sequentially with water, saturated aqueous NaHCO3 and brine, and dried over anhydrous MgSO4. After filtration, the filtrate was concentrated under reduced pressure and the residue was purified by silica gel column chromatography (eluent: hexane/ethyl acetate = 95/5 to 80/20) to afford 5-(benzyloxy)-2-bromobenzaldehyde as a colorless oil (140 mg, 97% yield).
References
[1] Bulletin of the Korean Chemical Society, 2010, vol. 31, # 7, p. 2047 - 2050
[2] Patent: EP2123637, 2009, A1. Location in patent: Page/Page column 110
[3] Patent: US2011/82295, 2011, A1. Location in patent: Page/Page column 14
[4] Journal of Organic Chemistry, 2016, vol. 81, # 20, p. 10062 - 10070
[5] Journal of Organic Chemistry, 1999, vol. 64, # 13, p. 4949 - 4952