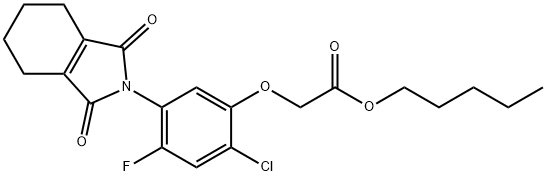
Chemical Properties
Pure product is white powdery solid. Melting point 88.9~90.1℃, relative density 1.33(20℃), vapor pressure 1.0×10-5Pa (25℃), partition coefficient 4.99 (20℃), solubility at 25℃: methanol 47.8g/L, n-octanol 16.0g/L, acetone 590g/L, n-hexane 3.28g/L, water 0.189mg/L, half-life in water 6min (pH=9), 19h (pH=7), 42d (pH=5). 6min (pH=9), 19h (pH=7), 42d (pH=5).
Uses
Flumiclorac-pentyl is a herbicide.
Definition
ChEBI: Flumiclorac pentyl is a pyrroline.
Synthesis
Flumiclorac-pentyl is produced via a 5–6 step synthesis that commences with the preparation of the hexahydrophthalic anhydride core through Diels-Alder cycloaddition of maleic anhydride with cis-1,3-butadiene under high-pressure conditions in benzene, followed by ring opening to the diacid and condensation with methylamine in water to form N-methyl-hexahydrophthalimide. This imide is then lithiated and coupled via nucleophilic substitution to 2-chloro-4-fluoro-5-nitrophenyl acetate. Reduction of the nitro group using yields the amine intermediate, which undergoes intramolecular cyclisation with the acetate to form the cyclic imide.
Metabolic pathway
In the feces and urine of rats, more than 90% of
administered 14C-flumiclorac pentyl is excreted within
48 h of treatment. Flumiclorac pentyl is rapidly and
extensively metabolized. The major fecal metabolites
are identified as the sulfonic acid conjugates, and the
major urinary metabolite is identified as 5-amino-2-
chloro-4-fluorophenoxyacetic acid. The metabolic
reactions include cleavage of the ester linkage, cleavage
of the imide linkage, hydroxylation and following
reduction at the cyclohexene ring of the cyclohexene-
1,2-dicarboxylic acid moiety, and incorporation of a
sulfonic acid group into the carbon carbon double bond
of the 3,4,5,6-tetrahydrophthalimide moiety.