Synthesis
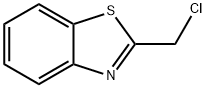
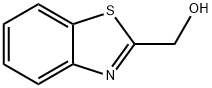
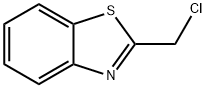
General procedure for the synthesis of 2-(chloromethyl)-1,3-benzothiazole from 2-hydroxymethylbenzothiazole: To a stirred solution containing 2-hydroxymethylbenzothiazole (2.5 g, 15.1 mmol), triphenylphosphine (4.89 g, 18.6 mmol), and anhydrous toluene (50 ml) was added, protected by nitrogen, carbon tetrachloride (15.0 ml, 155 mmol ). The reaction mixture was heated to reflux for 5 hours. After completion of the reaction, the reaction mixture was concentrated using a rotary evaporator to give the crude product. The crude product was purified by rapid chromatography on silica gel to afford 2-(chloromethyl)-1,3-benzothiazole (2.36 g, 85% yield) using 100% dichloromethane as eluent. The product was confirmed by mass spectrometry (ion spray): m/z = 183.9 (M + 1); 1H NMR (CDCl3) data were as follows: δ= 8.04-8.01 (1H, m), 7.91-7.89 (1H, m), 7.53-7.49 (1H, m), 7.44-7.40 (1H, m), 4.95 (2H, s).
References
[1] Tetrahedron, 1988, vol. 44, # 7, p. 2021 - 2032
[2] Chemical Communications, 2017, vol. 53, # 54, p. 7545 - 7548
[3] Patent: WO2005/92885, 2005, A1. Location in patent: Page/Page column 55
[4] Zhurnal Obshchei Khimii, 1954, vol. 24, p. 1664,1667; engl. Ausg. 1645, 1647
[5] Zhurnal Obshchei Khimii, 1956, vol. 26, p. 797,798; engl. Ausg. 911, 912