Synthesis
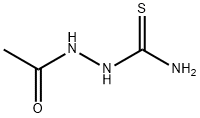
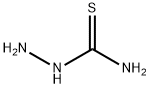
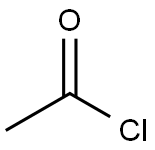
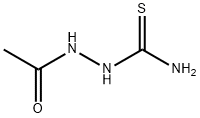
General procedure for the synthesis of acetylthiosemicarbazone from aminothiourea and acetyl chloride: preparation of the intermediate 2-acetylcarbazinethioformamide; Acetyl chloride (8.64 g, 0.11 mol) was added slowly and dropwise to an anhydrous tetrahydrofuran (THF, 100 mL) solution of aminothiourea (9.10 g, 0.10 mol) at 0 °C. The reaction mixture was heated to reflux and maintained for 3 hours. Upon completion of the reaction, it was cooled to room temperature and the reaction was quenched by the addition of water (100 mL) followed by extraction with ethyl acetate (3 x 100 mL). The organic phases were combined, washed with saturated saline, dried over anhydrous sodium sulfate, and concentrated under reduced pressure to remove the solvent. The crude product was purified by column chromatography (eluent: methanol/dichloromethane=1/1, v/v) to afford the target compound acetylthiosemicarbazone (8.64 g, 65% yield). The results of mass spectrometry (electrospray ionization, ESI) analysis: m/z calculated value C3H7N3OS [M+H]+ 133.03, measured value 134.0 (MH+).
References
[1] Patent: WO2009/87220, 2009, A1. Location in patent: Page/Page column 57
[2] Yakugaku Zasshi, 1952, vol. 72, p. 376
[3] Chem.Abstr., 1953, p. 3856
[4] Synthetic Communications, 2004, vol. 34, # 22, p. 4189 - 4198
[5] Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy, 2010, vol. 76, # 5, p. 531 - 536