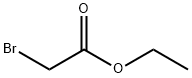
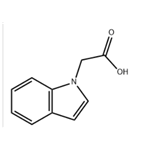
Uses
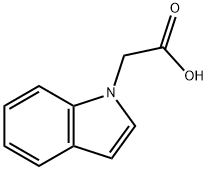
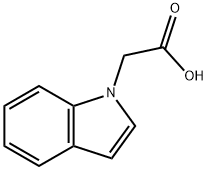
Indol-1-yl-acetic Acid is a reactant used in the preparation of conformationally restrained analogs of pravadoline; nanomolar potent, enantioselective, (aminoalkyl)indole agonists of the cannabinoid receptor.
Synthesis
Step 2: Preparation of 2-(1H-indol-1-yl)acetic acid
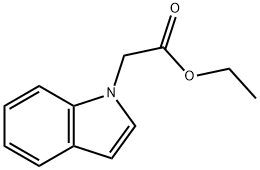
Ethyl 2-(1H-indol-1-yl)acetate (2.03 g, 10 mmol) was dissolved in ethanol (40 mL), aqueous sodium hydroxide solution (1 N, 50 mL) was added, and the reaction was stirred at room temperature for 1.5 hours. Upon completion of the reaction, the reaction solution was adjusted to slightly acidic with dilute hydrochloric acid and the mixture was subsequently concentrated under reduced pressure. The residue was extracted with ethyl acetate and the organic phase was washed with brine and the solvent was removed under reduced pressure to give pure 2-(1H-indol-1-yl)acetic acid (1.6 g, 87% yield), (LC/MS: m/z = 176.3 [M+H]+).
References
[1] Patent: WO2015/106025, 2015, A1. Location in patent: Paragraph 0334
[2] Patent: US2016/326123, 2016, A1. Location in patent: Paragraph 0546
[3] Journal of Heterocyclic Chemistry, 1984, vol. 21, p. 975 - 976
[4] European Journal of Medicinal Chemistry, 1989, vol. 24, p. 145 - 154
[5] Patent: WO2006/55725, 2006, A2. Location in patent: Page/Page column 188-189