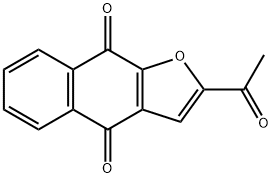
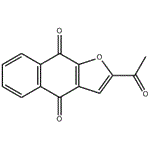
Description
Napabucasin is an inhibitor of cancer cell stemness. It reduces growth in a panel of cancer cells enriched for high stemness (IC
50s = 0.291-1.249 μM) and decreases protein and gene expression of various self-renewal and pro-survival markers including Nanog, Smo, Axl, Atm, and Bmi-1 in a concentration-dependent manner in FaDu head and neck squamous cell carcinoma (HNSCC) cells.
In vivo, napabucasin (20 mg/kg per day) inhibits tumor growth in a PaCa-2 mouse xenograft model and prevents tumor regrowth after cessation of treatment. It blocks formation of spleen and liver metastases in an HT29 intrasplenic nude mouse model system (ISMS) model. Napabucasin (40 mg/kg, i.p.) also reduces tumor volume in PC3 and 22RV1 prostate cancer mouse xenograft models.
Uses
Napabucasin is a cancer cell stemness inhibitor which attenuates the progression of prostate cancer.
in vivo
Napabucasin (40 mg/kg) or Docetaxel significantly reduces xenograft tumor growth and tumor volume (TV) compared with PBS (P<0.05). Notably, while no differences are observed between the Napabucasin and the docetaxel groups in PC-3 mouse xenograft models, the TV in Napabucasin group is even lower than docetaxel group in 22RV1 mouse xenograft models (P<0.05). Additionally, Napabucasin or docetaxel also significantly reduces tumor weight compared with PBS (P<0.05)[1].
References
[1] YOUZHI LI. Suppression of cancer relapse and metastasis by inhibiting cancer stemness.[J]. Proceedings of the National Academy of Sciences of the United States of America, 2015: 1839-1844. DOI:
10.1073/pnas.1424171112[2] JOLEEN M HUBBARD Axel G. Napabucasin: An Update on the First-in-Class Cancer Stemness Inhibitor.[J]. Drugs, 2017, 77 10: 1091-1103. DOI:
10.1007/s40265-017-0759-4[3] YIMING ZHANG. Suppression of prostate cancer progression by cancer cell stemness inhibitor napabucasin[J]. Cancer Medicine, 2016, 5 6: 1251-1258. DOI:
10.1002/cam4.675[4] LAUREN MACDONAGH . BBI608 inhibits cancer stemness and reverses cisplatin resistance in NSCLC[J]. Cancer letters, 2018, 428: Pages 117-126. DOI:
10.1016/j.canlet.2018.04.008