Synthesis
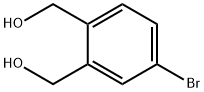
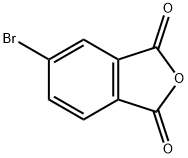
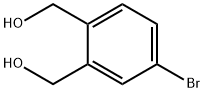
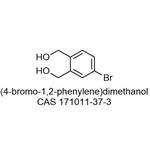
General procedure for the synthesis of 4-bromo-2-(hydroxymethyl)benzyl alcohol from 5-bromoisobenzofuran-1,3-dione: To an anhydrous toluene solution of 5-bromoisobenzofuran-1,3-dione (106 mg, 0.47 mmol) was slowly added a toluene solution of 1 M diisobutylaluminum hydride (DIBAL-H) (3 mL, 3 mmol) under ice-bath cooling conditions. The reaction mixture was stirred at room temperature for 1.5 hours. Subsequently, 10% aqueous hydrochloric acid solution (0.7 mL) was slowly added at 0°C. The reaction mixture was diluted with toluene (1 mL) and continued to be stirred at room temperature for 1 hour. The reaction mixture was filtered through diatomaceous earth and extracted with ethyl acetate (20 mL x 3). The organic phases were combined, washed with saturated saline (10 mL), dried over anhydrous magnesium sulfate and concentrated under reduced pressure. The residue was purified by silica gel column chromatography (eluent: hexane/ethyl acetate = 1:1) to afford the target product 4-bromo-2-(hydroxymethyl)benzyl alcohol (89 mg, 91% yield) as a colorless solid with a melting point of 67-67.5 °C.
References
[1] Heterocycles, 2009, vol. 77, # 2, p. 991 - 1005
[2] Synlett, 2013, vol. 24, # 19, p. 2510 - 2514
[3] Research on Chemical Intermediates, 2013, vol. 39, # 1, p. 139 - 146
[4] Patent: EP1659129, 2006, A1. Location in patent: Page/Page column 100
[5] Patent: WO2017/87854, 2017, A1. Location in patent: Page/Page column 00315