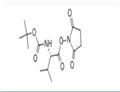
Chemical Properties
White powder
reaction suitability
reaction type: Boc solid-phase peptide synthesis
Synthesis
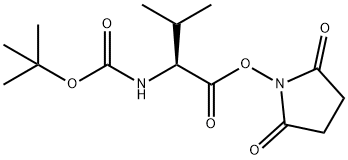
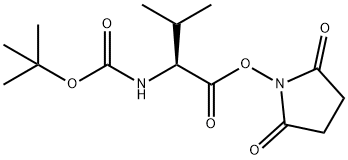
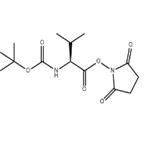
Step A: Boc-L-valine (1.03 g, 4.74 mmol), N-hydroxysuccinimide (1.22 g, 10.6 mmol), and 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDC, 1.60 g, 8.35 mmol) were sequentially added to a dry round-bottom flask. The above reagents were dissolved in anhydrous dichloromethane (30 mL), and the reaction system was sealed through a rubber septum and displaced three times with argon to exclude oxygen. Subsequently, the reaction mixture was stirred at room temperature.After 3 days, thin layer chromatography (TLC) detection (ninhydrin color development) confirmed that the Boc-L-valine was fully reacted. The reaction solution was washed sequentially with water, saturated aqueous sodium bicarbonate, and the aqueous phase was back-extracted with dichloromethane. The organic phases were combined, washed with saturated saline, dried over anhydrous sodium sulfate and filtered. The filtrate was concentrated under reduced pressure and dried under vacuum to obtain white solid Boc-L-valine hydroxysuccinimide ester (1.52 g, 100% yield). The product was characterized by 1H-NMR (300 MHz, CDCl3): δ 4.98 (broad peak, 1H), 4.58 (double peak, 1H), 2.82 (multiple peaks, 4H), 2.27 (multiple peaks, 1H), 1.44 (single peak, 9H), 1.03 (double peaks, 6H).
References
[1] Patent: WO2016/160615, 2016, A1. Location in patent: Paragraph 0444; 0445
[2] Journal of Medicinal Chemistry, 1998, vol. 41, # 14, p. 2461 - 2480
[3] Journal of Medicinal Chemistry, 1991, vol. 34, # 9, p. 2852 - 2857
[4] Journal of the Chemical Society, Dalton Transactions: Inorganic Chemistry (1972-1999), 1984, p. 2305 - 2308
[5] Journal of Organic Chemistry, 1986, vol. 51, # 24, p. 4580 - 4585