Synthesis
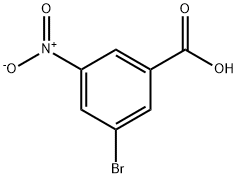
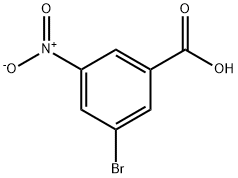
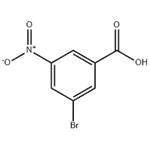
Part G: Preparation of 3-bromo-5-nitrobenzoic acid
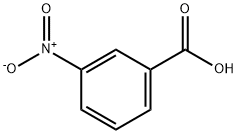
3-Nitrobenzoic acid (16.7 g, 100 mmol) was dissolved in a mixed solution of trifluoroacetic acid (50 mL) and sulfuric acid (20 mL) at 50 °C. Subsequently, N-bromosuccinimide (26.7 g, 150 mmol) was added in three batches over 3 hours. The reaction mixture was stirred at 50 °C for 16 h and then cooled to room temperature. The reaction mixture was slowly poured into ice water (200 mL) and extracted three times with ethyl acetate. The organic layers were combined, dried over anhydrous sodium sulfate and concentrated under reduced pressure. The residue was recrystallized by dichloromethane to give the white solid product 3-bromo-5-nitrobenzoic acid (17.7 g, 72% yield). The structure of the product was confirmed by 1H NMR (300 MHz, DMSO-d6): δ 13.8 (broad peak, 1H, carboxylic acid proton), 8.60 (single peak, 1H, aromatic proton), 8.55 (single peak, 1H, aromatic proton), 8.37 (single peak, 1H, aromatic proton).
References
[1] ChemMedChem, 2014, vol. 9, # 5, p. 973 - 983
[2] Journal of the American Chemical Society, 2017, vol. 139, # 36, p. 12579 - 12590
[3] Journal of Medicinal Chemistry, 2016, vol. 59, # 4, p. 1556 - 1564
[4] Journal of Organic Chemistry, 2007, vol. 72, # 15, p. 5867 - 5869
[5] Chemistry - A European Journal, 2011, vol. 17, # 10, p. 2987 - 2995