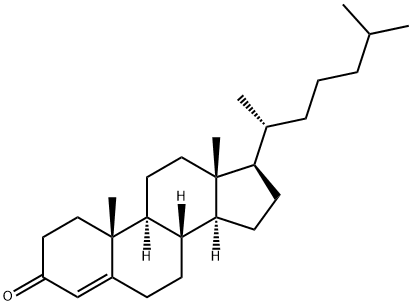
Description
Cholestanone is a cholesterol metabolite that has a keto group in place of the 3-hydroxy group on cholesterol. It decreases TGF-β-induced Smad2 phosphorylation and TGF-β expression and prevents inhibition of DNA synthesis by TGF-β in Mv1Lu cells when used at a concentration of 50 μg/ml. Increased fecal excretion of cholestenone is correlated with an increased risk of colorectal cancer. Cholestenone reduces serum cholesterol levels in a variety of animal models but is toxic to rats when administered at doses of 700-1,000 mg/kg per day, inducing hypertrophy in and reducing the activity of the adrenal gland. It has been used as synthetic intermediate in the synthesis of steroids.
References
[1] G SALEN. Inverse relationship between plasma cholestanol concentrations and bile acid synthesis in sitosterolemia.[J]. Journal of Lipid Research, 1994, 35 10: 1878-1887.
[2] CHUN-LIN CHEN. Cholest-4-en-3-one attenuates TGF-β responsiveness by inducing TGF-β receptors degradation in Mv1Lu cells and colorectal adenocarcinoma cells.[J]. Journal of Receptors and Signal Transduction, 2017, 37 2: 189-199. DOI:
10.1080/10799893.2016.1203944[3] D STEINBERG J A D S FREDRICKSON. Effects of 4-cholestenone in animals and in man.[J]. Proceedings of the Society for Experimental Biology and Medicine, 1958, 97 4: 784-790. DOI:
10.3181/00379727-97-23880[4] KE WU. Production, Purification, and Identification of Cholest-4-en-3-one Produced by Cholesterol Oxidase from Rhodococcus sp. in Aqueous/Organic Biphasic System.[J]. Biochemistry Insights, 2015, 8 Suppl 1: 1-8. DOI:
10.4137/bci.s21580