Synthesis
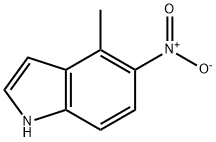
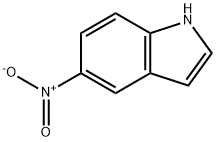
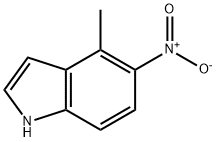
General procedure for the synthesis of 4-methyl-5-nitro-1H-indole using 5-nitroindole as starting material: to an anhydrous THF solution (20 mL) of 5-nitroindole (purchased from Aldrich, 0.82 g, 5.1 mmol) was slowly added methylmagnesium bromide (5.0 mL, 15 mmol), and the reaction was stirred continuously for 1 hr at 25 °C. Subsequently, the reaction was quenched by addition of THF solution of tetrachloro-1,4-benzoquinone (1.2 g, 4.9 mmol). The reaction mixture was concentrated under reduced pressure to give a dark colored solid. This solid was purified by column chromatography (eluent: 30% ethyl acetate/hexane) to give the final target product 4-methyl-5-nitro-1H-indole (0.72 g, 4.1 mmol, 80% yield).