Description
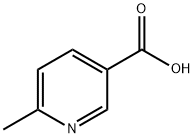
6-Methylnicotinic acid is a biochemical reagent that can be used as a pharmaceutical intermediate or raw material for organic synthesis. 6-Methylnicotinic acid is used as the starting material and 6-methylnicotine can be synthesized through four steps of esterification, condensation, ring-opening rearrangement, and reduction. As an alkaloid, 6-methylnicotine has similarities with nicotine, but slight changes in its molecular structure may lead to differences in its pharmacological effects and addictiveness. It may be used in products such as electronic cigarettes as a substitute or supplement for nicotine.
Chemical Properties
6-Methylnicotinic acid is a white to light yellow to light orange powder to crystals with a melting point of 210 °C. Store under inert gas at room temperature.
Uses
6-Methylnicotinic acid is an intermediate of the drug etoricoxib (a non- steroidal anti-inflammatory drug for the treatment of arthritis and osteoarthritis). It is also used as organic intermediates.
Uses
A degradation product of Nicotinic acid (N429250). An impurity of Nicotinic acid.
General Description
The co-ordination complex of the formula [Ni(L1)
2(H
2O)
4].4H
2O where L1H = 6-methylpyridine-3-carboxylic acid was structurally charaterized using X-ray single crystal analysis.
Synthesis
6-Methylnicotinic acid is prepared by selective oxidation of 2-methyl-5-alkylpyridine, wherein the alkyl group has 2 to 8 carbon atoms, using nitric acid at a concentration of 70 to 100 weight percent HNO3, wherein 6 to 15 moles of NHO3 are used per mole of pyridine, and the reaction is carried out at a temperature range of 250 to 325*F. The reaction time is 10 to 60 minutes, and superatmospheric pressure is used to effectively maintain the reactants in the liquid phase, and 6-methylnicotinic acid is recovered from the resulting reaction mixture.