Synthesis
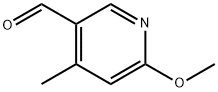
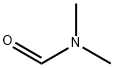
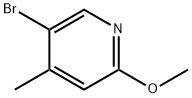
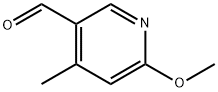
General procedure for the synthesis of 5-formyl-2-methoxy-4-methyl-4-methylpyridine from 2-methoxy-4-methyl-5-bromopyridine and N,N-dimethylformamide: 5-bromo-2-methoxy-4-methylpyridine (2.6 g, 13 mmol) was dissolved in anhydrous THF (40 mL) and cooled to -70 °C under argon protection. A hexane solution of 1.6 M n-butyllithium (8.1 mL, 14 mmol) was slowly added over 10 min with stirring. The reaction temperature was maintained at -70 °C and stirring was continued for 30 min. Subsequently, anhydrous DMF (1.2 mL, 15 mmol) was added in batches, keeping the temperature constant. After addition, stirring was continued at -70 °C for 30 min, then brought to room temperature and stirred overnight. Upon completion of the reaction, the reaction was quenched with 1 M hydrochloric acid (40 mL) and subsequently extracted three times with tert-butyl methyl ether (TBME). The organic phases were combined, washed with brine, dried over anhydrous sodium sulfate, filtered and concentrated. Purification by column chromatography using tert-butyl methyl ether-light petroleum ether (1:1) as eluent afforded 1.8 g (91% yield) of 5-formyl-2-methoxy-4-methylpyridine as a light yellow solid.LC-MS m/z 152 (M + 1); 1H NMR (CDCl3) δ 10.1 (s, 1H), 8.55 (s, 1H), 6.61 ( s, 1H), 4.05 (s, 3H), 2.60 (s, 3H) ppm.
References
[1] Patent: WO2006/65215, 2006, A1. Location in patent: Page/Page column 19; 35
[2] Synthetic Communications, 2009, vol. 39, # 17, p. 3060 - 3068
[3] Patent: WO2015/158427, 2015, A1. Location in patent: Page/Page column 84; 85