Chemical Properties
White Crystal
Uses
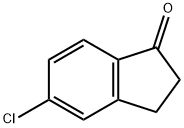
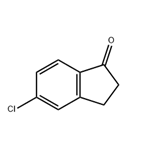
5-Chloro-1-indanone may be used as starting reagent for the preparation of 5-chloro-2-methoxycarbonyl-1-indanone. It may also be used for the preparation of important biomedical compounds such as anticonvulsants, anticholinergics and diarylsulfonylureas, having potential activity against solid tumors. It participates in the Irie?s synthesis of substituted pyridines.
Uses
5-Chloro-1-indanone is the important intermediate of du pont company's new varieties of pesticides indoxacarb (popular name: indoxacarb), is also a kind of important medicine intermediate simultaneously.
5-Chloro-1-indanone is a 5-halo-1-indanone. It participates in the Irie′s synthesis of substituted pyridines. 5-Chloro-1-indanone has a stable triclinic crystal structure and has intermolecular forces of C-H...O, C-H...Π, CO...Cl and Π...Π types.
5-Chloro-1-indanone may be used as starting reagent for the preparation of 5-chloro-2-methoxycarbonyl-1-indanone. It may be used for the preparation of important biomedical compounds such as anticonvulsants, anticholinergics and diarylsulfonylureas, having potential activity against solid tumors.
Preparation
3-chlorobenzaldehyde as raw material first reacts with propionic acid to prepare 3-chloro-phenylpropionic acid, which is then subjected to Friedel-Crafts acylation reaction to prepare 5-chloro-1-indanone. Organic solvents of formic acid diethylamine participate in the first step, and the reaction temperature is 20-150℃. An organic solvent of methylene chloride and a catalyst of zinc chloride participate in the second step, and the reaction temperature is -10 to 80 ℃.
Synthetic method of 5-chloro-1-indanone
General Description
5-Chloro-1-indanone is a 5-halo-1-indanone. It participates in the Irie′s synthesis of substituted pyridines. 5-Chloro-1-indanone has a stable triclinic crystal structure and has intermolecular forces of C-H...O, C-H...Π, CO...Cl and Π...Π types.