Description
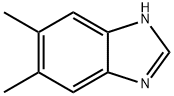
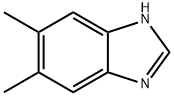
5,6-Dimethylbenzimidazole is a dimethylbenzimidazole that has been found in S. meliloti and is an intermediate in the biosynthesis of the corrin macrocycle vitamin B12 (Item No. 18425).1 It is formed from the flavin coenzyme riboflavin 5ʹ-monophosphate (Item No. 18167) by the oxidoreductase BluB in S. meliloti. 5,6-Dimethylbenzimidazole forms the axial region of vitamin B12 and chelates the central cobalt.WARNING This product is not for human or veterinary use.
Chemical Properties
light beige powder
Definition
ChEBI: A dimethylbenzimidazole carrying methyl substituents at positions 5 and 6.
Synthesis
To a 16 mL reaction flask equipped with a PTFE/silicone septum and a nitrogen bubbler was added N-benzylbenzimidazole (208.1 mg, 1.0 mmol), dried Pd/C catalyst (10 wt%, 20 mg) and THF (5 mL). Triethylsilane (320 μL, 2.0 mmol) was added dropwise to the mixture under nitrogen protection, followed by continuous stirring at room temperature for 14 hours. After completion of the reaction, the reaction mixture was filtered through a 0.45 μm PTFE syringe filter and the filtrate was concentrated under reduced pressure. The residue was purified by column chromatography (eluent: 0 to 10% methanol/dichloromethane gradient) to afford 5,6-dimethylbenzimidazole as a colorless solid (117.6 mg, 0.996 mmol, 99% yield). The reaction typically exhibits an induction period of 5 to 30 min, which can be observed by the phenomenon of gas release (i.e., bulging) from the reaction mixture. The use of a dry Pd/C catalyst is essential; wet Pd/C results in lower yields or the reaction does not proceed.
Purification Methods
Crystallise 5,6-dimethylbenzimidazole from diethyl ether. It sublimes at 140o/3mm. [Beilstein 23/6 V 454.]
References
[1] Tetrahedron Letters, 2015, vol. 56, # 21, p. 2688 - 2690