Synthesis
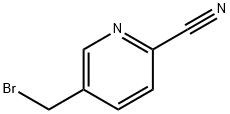
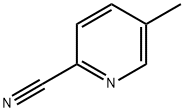
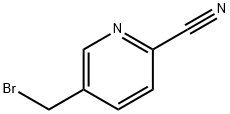
General procedure for the synthesis of 5-(bromomethyl)-2-cyanopyridine using 2-cyano-5-methylpyridine as starting material: commercially available 2-cyano-5-methylpyridine (1.90 g, 16.1 mmol) was dissolved in chloroform (100 mL), followed by the addition of N-bromosuccinimide (4.29 g, 24.1 mmol) and α,α-azobisisobutyronitrile (0.792 g, 4.82 mmol). The reaction mixture was stirred at 60 °C for 6 hours. After completion of the reaction, the mixture was cooled in an ice bath, the precipitate was separated by filtration and the filtrate was concentrated under reduced pressure. The resulting residue was purified by silica gel column chromatography to afford the target compound 5-(bromomethyl)-2-cyanopyridine (1.69 g, 53% yield).1H NMR (CDCl3) δ (ppm): 4.80 (s, 2H), 8.03 (dd, J = 0.92, 8.07 Hz, 1H), 8.12 (dd, J = 2.20, 8.07 Hz. 1H), 8.81 (d, J = 1.83 Hz, 1H).
References
[1] Patent: EP2163554, 2010, A1. Location in patent: Page/Page column 52-53
[2] Patent: WO2007/28083, 2007, A2. Location in patent: Page/Page column 69-70
[3] Bioorganic and Medicinal Chemistry Letters, 2002, vol. 12, # 7, p. 1017 - 1022
[4] Journal of Medicinal Chemistry, 2003, vol. 46, # 17, p. 3612 - 3622
[5] Patent: WO2011/20615, 2011, A1. Location in patent: Page/Page column 57; 58