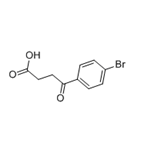
Synthesis
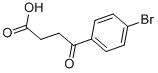
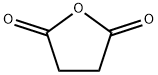
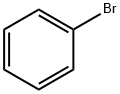
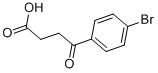
The general procedure for the synthesis of 3-(4-bromobenzoyl)propionic acid from butanedioic anhydride and bromobenzene was as follows: butanedioic anhydride (5.0 g, 50 mmol) and bromobenzene (48 g, 300 mmol) were cooled to 0 °C. Anhydrous aluminum chloride (13.3 g, 100 mmol) was added to the reaction mixture under nitrogen protection and stirred at 0 °C for 4 hours. Subsequently, the reaction mixture was slowly warmed to room temperature and stirring was continued for 96 hours under nitrogen protection. Upon completion of the reaction, the mixture was cooled again to 0 °C, concentrated hydrochloric acid (125 mL) was slowly added and stirred under nitrogen protection for 1 hour. The reaction mixture was filtered and washed with water (1 L) to give a light yellow solid. The solid was purified by recrystallization from toluene to give 4-(4-bromophenyl)-4-oxobutanoic acid (12 g, 46.68 mmol, 93.4% yield). Mass spectrometry (ESI-quadrupole) m/z: calculated values of C10H9BrO3 were 255.97, 257.97; measured values: 258.0 (11), 257.0 (98), 256.0 (12), 255.0 (100), 213.0 (18), 211.0 (17) (in negative ion mode). High performance liquid chromatography (HPLC) retention time: 24.4 min. 1H NMR (500 MHz, DMSO-d6) δ: 2.59 (2H, triple peaks, J=6.5 Hz, CH2), 3.21 (2H, triple peaks, J=6.5 Hz, CH2), 7.88 (2H, double peaks, J=8.8 Hz, ArH), 7.96 (2H. double peak, J=8.8Hz, ArH), 12.19 (1H, broad single peak, OH).
References
[1] Molecular Crystals and Liquid Crystals Science and Technology Section A: Molecular Crystals and Liquid Crystals, 2001, vol. 365, p. 181 - 188
[2] Journal of Organic Chemistry, 2001, vol. 66, # 22, p. 7283 - 7286
[3] Organic Syntheses, 2002, vol. 79, p. 204 - 204
[4] Bioorganic and Medicinal Chemistry, 2009, vol. 17, # 13, p. 4345 - 4359
[5] Organic Process Research and Development, 2014, vol. 18, # 1, p. 215 - 227