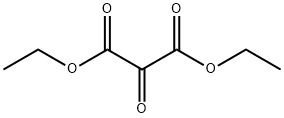
Chemical Properties
Clear yellow liquid
Uses
Diethyl ketomalonate, reagent employed in Wittig and Aza-Wittig reactions for synthesis of triazoles, and 2-azadienes, respectively.
Application
Diethyl ketomalonate can be used in electron-deficient heterodienophile; Diels-Alder reaction; heterodipolarophile; [3 + 2] cycloaddition; 1,3-diyl trapping agent; 1,4-dipolar cycloaddition; [2 + 2] cycloaddition; enophile; ene reaction; electrophilic addition; Wittig reactions; condensation with amines; Pictet-Spengler reactions.
Preparation
Diethyl ketomalonate can be prepared by bromination of Diethyl Malonate to the dibromomalonic ester, exchange of one bromine atom for acetate and α-elimination of acetyl bromide. Alternatively, diethyl malonate is condensed with benzaldehyde to afford the Knoevenagel product which is cleaved by ozonolysis.
Synthesis
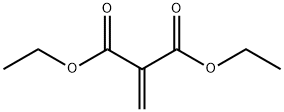
The general procedure for the synthesis of diethyl ketomalonate from diethyl methylidenemalonate was as follows: 258 g of diethyl methylidenemalonate prepared by Knoevenagel condensation reaction of malonic acid and formaldehyde was dissolved in 1 liter of methanol, followed by ozonation reaction. Upon completion of the reaction, 5 g of 10% Pd-on-C was added as a hydrogenation catalyst and placed in 200 ml of methanol to carry out the hydrogenation reaction, with a hydrogen consumption of 28.9 standard liters (86% of the theoretical value was reached). The reaction mixture was post-treated according to the method of Example 5 and subsequently concentrated under vacuum to give 214 g of diethyl ketomalonate with a boiling point of 10.0-112°C/20 mmHg in 82% yield of the theoretical value.
storage
Diethyl ketomalonate is stable for at least 6 months if stored in the refrigerator.
References
[1] Patent: US4769464, 1988, A
[2] Tetrahedron Letters, 1981, vol. 22, # 46, p. 4607 - 4610