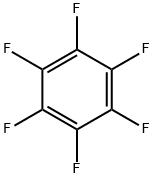
Hexafluorbenzol
Bezeichnung:Hexafluorbenzol
CAS-Nr392-56-3
Englisch Name:hexafluorobenzene
CBNumberCB7152315
SummenformelC6F6
Molgewicht186.05
MOL-Datei392-56-3.mol
Synonyma
Hexafluorbenzol
Hexafluorbenzol physikalisch-chemischer Eigenschaften
| Schmelzpunkt | 3.7-4.1 °C (lit.) |
| Siedepunkt | 80-82 °C (lit.) |
| Dichte | 1.612 g/mL at 25 °C (lit.) |
| Brechungsindex | n |
| Flammpunkt | 50 °F |
| storage temp. | Sealed in dry,Room Temperature |
| Aggregatzustand | Liquid |
| Farbe | Clear colorless to slightly yellow |
| Wichte | 1.612 |
| Wasserlöslichkeit | Immiscible with water. |
| Merck | 14,4686 |
| BRN | 1683438 |
| Dielectric constant | 2.0499999999999998 |
| Stabilität | Stable. Incompatible with strong oxidizing agents. May form complexes with transition metals which can explode when heated. Highly flammable. |
| CAS Datenbank | 392-56-3(CAS DataBase Reference) |
| NIST chemische Informationen | Benzene, hexafluoro-(392-56-3) |
| EPA chemische Informationen | Benzene, hexafluoro- (392-56-3) |
| Kennzeichnung gefährlicher | F,Xi |
| R-Sätze: | 11-36/37/38 |
| S-Sätze: | 16-33-7/9-29-26-37/39 |
| RIDADR | UN 1993 3/PG 2 |
| WGK Germany | 3 |
| RTECS-Nr. | DA3050000 |
| F | 10 |
| Hazard Note | Highly Flammable |
| TSCA | Yes |
| HazardClass | 3 |
| PackingGroup | II |
| HS Code | 29039990 |
| Toxizität | guinea pig,LCLo,inhalation,11800ppm/4H (11800ppm),BEHAVIORAL: ATAXIASENSE ORGANS AND SPECIAL SENSES: LACRIMATION: EYELUNGS, THORAX, OR RESPIRATION: OTHER CHANGES,National Technical Information Service. Vol. OTS0571186, |

