Medical Uses and Side Effect of Clobetasol Propionate
Patents & Indications
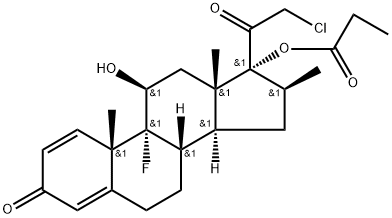
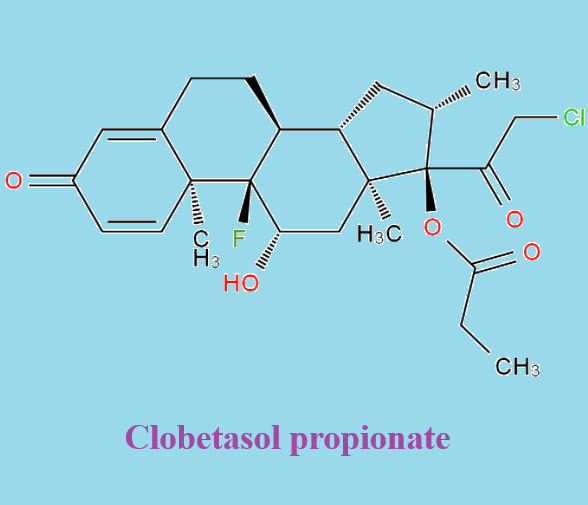
Clobetasol propionate is the 17-O-propionate ester of clobetasol and it derives from a clobetasol and a propionic acid. A potent corticosteroid, it is used to treat various skin disorders, including exzema and psoriasis.
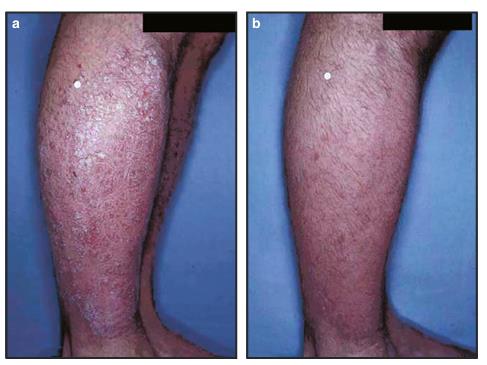
Clobetasol propionate was patented in 1968 and came into medical use in 1978. It is available as a generic medication. Clobetasol propionate is used for the treatment of various skin disorders including eczema (figure 1), herpes labialis, psoriasis, and lichen sclerosus. It is also used to treat several auto-immune diseases, including alopecia areata, lichen planus (auto immune skin nodules), and mycosis fungoides (T-cell skin lymphoma). It is used as first-line treatment for both acute and chronic GVHD of the skin [2].
Figure 1 Cream for skin treatment with clobetasol propionate
Clobetasol Propionate is the propionate salt form of clobetasol, a topical synthetic corticosteroid with anti-inflammatory, anti-pruritic, and vasoconstrictive properties. Clobetasol propionate exerts its effect by binding to cytoplasmic glucocorticoid receptors and subsequently activates glucocorticoid receptor mediated gene expression. This results in the synthesis of certain anti-inflammatory proteins, while inhibiting the synthesis of certain inflammatory mediators. Specifically, clobetasol propionate appears to induce phospholipase A2 inhibitory proteins, thereby controlling the release of the inflammatory precursor arachidonic acid from membrane phospholipids by phospholipase A2 [1].
Potential Side Effects
Use clobetasol propionate on the skin only but do not use it on the face, groin, or underarms unless directed to do so by your doctor because there are some side effects, such as: burning, stinging, itching, dryness, or redness at the application site may occur when first applied to the skin, but many people using this medication do not have serious side effects. Rarely, it is possible this medication will be absorbed from the skin into the bloodstream. Cushing's syndrome has been reported in infants and adults as a result of prolonged use of topical clobetasol propionate formulations. The following additional local adverse reactions have been reported with topical corticosteroids, and they may occur more frequently with the use of occlusive dressings and higher potency corticosteroids. These reactions are listed in an approximately decreasing order of occurrence: dryness, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, irritation, striae, and miliaria [3]. A very serious allergic reaction to this drug is rare.
Clobetasol propionate has not been tested for teratogenicity when applied topically; however, it is absorbed percutaneously, and when administered subcutaneously it was a significant teratogen in both the rabbit and mouse. Clobetasol propionate has greater teratogenic potential than steroids that are less potent. Teratogenicity studies in mice using the subcutaneous route resulted in fetotoxicity at the highest dose tested (1 mg/kg) and teratogenicity at all dose levels tested down to 0.03 mg/kg. These doses are approximately 1.4 and 0.04 times, respectively, the human topical dose of clobetasol propionate gel, cream and ointment. Abnormalities seen included cleft palate and skeletal abnormalities. In rabbits, clobetasol propionate was teratogenic at doses of 3 and 10 mcg/kg. These doses are approximately 0.02 and 0.05 times, respectively, the human topical dose of clobetasol propionate gel, cream and ointment. Abnormalities seen included cleft palate, cranioschisis, and other skeletal abnormalities. There are no adequate and well-controlled studies of the teratogenic potential of clobetasol propionate in pregnant women.
References
[1] https://pubchem.ncbi.nlm.nih.gov/compound/Clobetasol-propionate
[2] https://en.wikipedia.org/wiki/Clobetasol_propionate
[3]https://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?setid=4a761afb-a48a-4a5a-8700-ad93601a260f&type=display
Related articles And Qustion
Lastest Price from Clobetasol propionate manufacturers

US $5.00-0.50/KG2025-05-06
- CAS:
- 25122-46-7
- Min. Order:
- 1KG
- Purity:
- 99% hplc
- Supply Ability:
- 500TONS

US $0.00-0.00/g2025-04-27
- CAS:
- 25122-46-7
- Min. Order:
- 10g
- Purity:
- 97%~102%,USP38
- Supply Ability:
- 50kg