Methane is a colorless, odorless, flammable hydrocarbon gas that is the simplest alkane. The root word, met, in methane is derived from the Greek root word methe meaning wine. Methylene was used in the early 19th century as the name for methanol, which is wood alcohol, CH3OH. Methylene comes from methe + hydē, the latter being the Greek word for wood, so methylene would mean wine from wood. Methanol got the names methylene and wood alcohol because it was discovered by Robert Boyle (1627–1691) in the 17th century by the destruction distillation of wood. Destructive distillation involves heating in the absence of air.
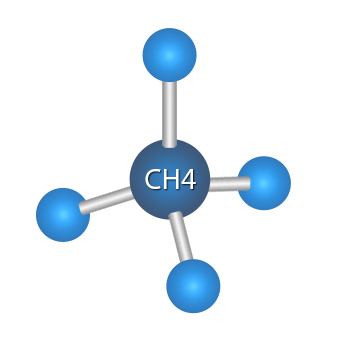
Methane is the first alkane and carries the suffix“ane” denoting an alkane, thus methe z + ane = methane. The carbon is at the center of the tetrahedron, which can be assumed to be an equilateral pyramid, with a hydrogen atom at each of the four corners of the tetrahedron.
Methane is the principal component of natural gas, with most sources containing at least 75% methane. Methane production occurs naturally through a process called methanogenesis. Methanogenesis involves anaerobic respiration by single-cell microbes collectively called methanogens.