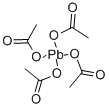
Lead(IV) acetate is an important oxidizing agent and a source of acetyloxy group used in organic synthesis. For example, 1,4-dioxene is prepared from dioxane involving 2-acetoxy-1,4-dioxane as an intermediate. Similarly, it is used for the preparation of bis(trifluoromethyl)diazomethane from hexafluoroacetone hydrazone. It also reacts with alkenes, alcohols having a delta-proton and di-n-butyl d-tartrate to get gamma-lactones, cyclic ethers and n-butyl glyoxylate respectively. It induces the cleavage of 1,2-diols to the corresponding aldehydes or ketones. It is actively involved in the Kochi reaction for the decarboxylation of carboxylic acids to alkyl halides and used as an alternative reagent to bromine in the Hofmann rearrangement.