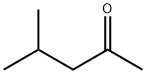
Products and mechanisms of the reaction between 4-methyl-2-pentanone and OH free radicals
What is 4-methyl-2-pentanone?
4-methyl-2-pentanone (also known as methyl isobutyl ketone (MIBK)) is an organic ketone compound that is widely found in daily foods or fruits, such as nuts, ginger, citrus and animal foods. In the MIBK structure, a carbonyl group is combined with two carbon atoms R2C=O (neither R can be a hydrogen atom). Ketones with one or more α-hydrogen atoms will undergo keto-enol isomerism, and the isomer is an enol. MIBK is also one of the top ten most commonly used organic solvents in industry and is often used as a solvent or intermediate in the chemical industry.
Products of the reaction of 4-methyl-2-pentanone with OH radical
Experimental conditions and products formation for the reaction of 4M2P with OH radical at T = 296 ± 2 K and atmospheric pressure.
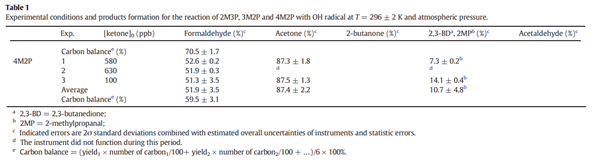
As shown in Table 1, the main products of the reaction of OH radical with 4M2P are acetone, 2-methylpropanal and formaldehyde, with yields of 87.4 ± 2.2%, 10.7 ± 4.8% and 51.9 ± 3.5%, respectively.
Reaction mechanism
The reaction of OH with ketones proceeds through initial H-atom abstraction from CH3-,-CH2- and/or >CH- groups depending on their position relative to the carbonyl group (>C=O) and the ketone chemical structure.
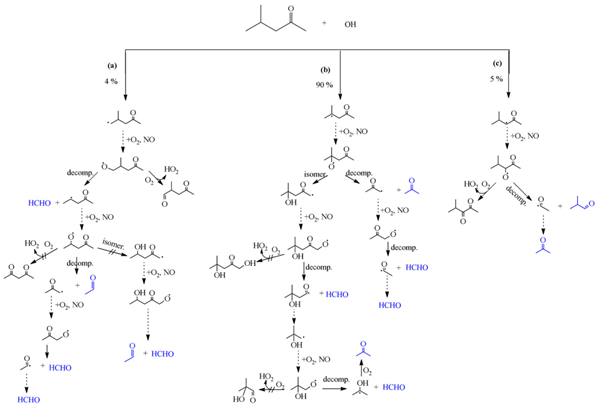
Fig. 6. (a–c): Reaction of OH radicals with 4M2P: proposed mechanism leading to the formation of observed reaction products. Compounds in blue represent products observed experimentally. The expected relative importance of the possible RO· reaction pathways are indicated by the arrows: → > ↛; The intermediary reaction of RO·/ RO2· is indicated by ⇢.
The observed products yields indicate that acetone and 2-methylpropanal must arise almost totally following H-abstraction from >CH- and -CH2- groups, respectively, as displayed in Fig. 6(a–c). Furthermore, the detected acetaldehyde using UHPLC-MS mainly formed from H-abstraction from -CH3. By taking account of the experimental formation yields of formaldehyde, acetone and 2-methylpropanal, the total carbon balance is estimated to be 59.5 ± 3.1%.
References:
[1] YANGANG REN. Atmospheric chemistry of ketones: Reaction of OH radicals with 2-methyl-3-pentanone, 3-methyl-2-pentanone and 4-methyl-2-pentanone.[J]. ACS Biomaterials Science & Engineering, 2021: 146249. DOI:10.1016/j.scitotenv.2021.146249.
Related articles And Qustion
See also
Lastest Price from 4-Methyl-2-pentanone manufacturers

US $1.00/KG2025-04-21
- CAS:
- 108-10-1
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt

US $0.00-0.00/kg2025-04-21
- CAS:
- 108-10-1
- Min. Order:
- 1kg
- Purity:
- 99.99%
- Supply Ability:
- 20 tons