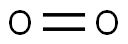Oxygen-Hazard and Toxicity
Description
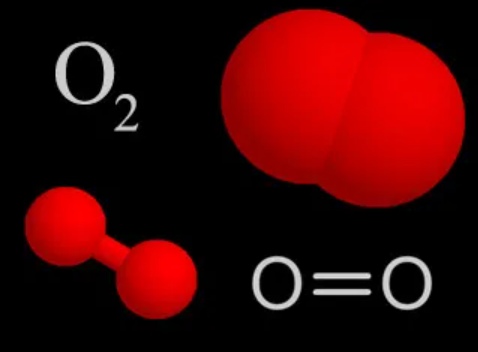
Oxygen, O2, is a colorless, tasteless, gaseous element essential to almost all forms of life. It promotes respiration and combustion. Oxygen comprises 20% of the earth's atmosphere and is the most abundant element in seawater and in the earth's crust. It is slightly soluble in water and alcohol, but combines readily with most other elements to form oxides. The electrolysis of water produces both oxygen and hydrogen.

Toxicity Data
OSHA recommends a minimum oxygen concentration of 19.5% for human occupancy.
Major Hazards
Powerful oxidizing agent; concentrations greater than 25% greatly enhance the combustion rate of many materials. Although oxygen itself is not flammable or explosive, as is sometimes believed, its mainhazard is that, in high concentrations, oxygen can cause other materials to burn much morerapidly.
Oxygen is toxic and deadly to breathe when in a pure state at elevated pressures. In addition,such pure oxygen promotes rapid combustion and can produce devastating fires, such asthe fire that killed the Apollo 1 crew on a test launch pad in 1967. It spread rapidly because thepure oxygen was at normal pressure rather than the one-third pressure used during flight.
Oxygen used for therapeutic purposes in adults can cause convulsions if the concentrationis too high. At one time, high levels of oxygen were given to premature infants to assist theirbreathing. It was soon discovered that a high concentration of O2 caused blindness in some ofthe infants. This practice has been abandoned, or the oxygen levels have since been reduced,and this is no longer a medical problem.
Toxicity
Oxygen is nontoxic under the usual conditions of laboratory use. Breathing pure oxygen at one atmosphere may produce cough and chest pains within 8 to 24 h, and concentrations of 60% may produce these symptoms in several days. Liquid oxygen can cause severe "burns" and tissue damage on contact with the skin due to extreme cold.
Flammability and Explosibility
Oxygen itself is nonflammable, but at concentrations greater than 25% supports and vigorously accelerates the combustion of flammable materials. Some materials (including metals) that are noncombustible in air will burn in the presence of oxygen.
Reactivity and Incompatibility
Oxygen is incompatible with combustible materials, including many lubricants and elastomers. Oil, greases, and other readily combustible substances should never be allowed to come in contact with O2 cylinders, valves, regulators, and fittings. Contact of liquid oxygen with many organic substances can lead to an explosion.
Accidents
In the event of skin or eye contact with liquid oxygen, seek medical attention for cryogenic burns. Do not enter areas of high oxygen gas concentration, which can saturate clothing and increase its flammability. Ventilate area to evaporate and disperse oxygen.
Disposal
Excess liquid oxygen should be allowed to evaporate in a well-ventilated outdoor area. Vent oxygen gas to outside location. Locations should be remote from work areas, open flames, or sources of ignition and combustibles. Return empty and excess cylinders of oxygen to manufacturer.