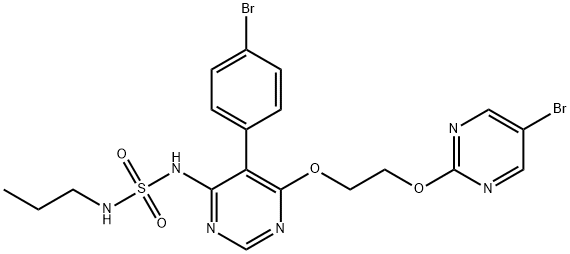
Macitentan: a dual endothelin-receptor antagonist
Macitentan is an orally active, non-peptide, potent dual ERA (ETA and ETB). In vitro, macitentan selectively inhibits the binding of endothelin-1 (ET-1) to ETA and ETB receptors. By inhibiting the effects of elevated ET-1 levels, ERAs reduce vasoconstriction, smooth muscle cell proliferation, and pulmonary vessel fibrosis. Macitentan is available as 10 mg film-coated tablets, and is to be taken orally at a dose of 10 mg once daily, with or without food. In Canada, macitentan is indicated for the long-term treatment of PAH (WHO Group I) to reduce morbidity in patients of WHO FC II or III whose PAH is either idiopathic, heritable, or associated with connective tissue disease or congenital heart disease.

Preparation of Macitentan
To a solution of THF (200.0 ml) and lithium amide (3.20 gm, 0.139), Λ/-5-(4- bromophenyl)-6-(-2-hydroxyethoxy)-4-pyrimidinyl-A/'-propylsulfamide (10.0 gm, 0.023 mol) was added at 0-5° C under nitrogen atmosphere and stirred the content at 0-5° C for 30 min, further DMF (40.0 ml) and 5- bromopyrimidin-2-yl 4-methylbenzenesulfonate (15.0 gm; 0.045 mol) was added to the reaction mass at 0-5° C. Stirred and heated reaction mass to 25-30°C and Maintain the reaction mass at 25-30° C for 24-30 hr. After completion of reaction monitored by HPLC, 5% w/v citric acid solution (250.0 ml) was added and the compound was extracted twice with ethyl acetate (150.0 ml x 2), followed by ethyl acetate layer was washed with 10% brine solution w/v (150.0 ml). Ethyl acetate layer was concentrated at 55-60°C under reduced pressure to produce /V-[5-(4-bromophenyl)-6-[2-[(5-bromo-2- pyrimidinyl)oxy]ethoxy]-4-pyrimidinyl]- v,-propyl sulfamide as a crude residue. The obtained residue was dissolved in methanol (240.0 ml) at 65-70 °C and decolorized by activated charcoal. The resulting solution was gradually cooled to room temperature then cooled to 0-5°C and maintained reaction mass at 0-5°C for 45-60 min. Obtained solid was filtered, washed with methanol (10.0 ml), suck dried to afford crude A/-[5-(4-bromophenylj-6-[2-[(5- bromo-2-pyrimidinyl)oxy]ethoxy]-4-pyrimidinyl]-//'-propylsulfamide [Macitentan]. [Yield = 7.0 gm; Purity (HPLC) 99.50%] [1]
Background and Hepatotoxicity
Macitentan (ma" si ten' tan) is a nonselective inhibitor of the endothelin-1 receptors, both type A and B. Inhibition of endothelin receptors disrupts the intracellular pathways that lead to vasoconstriction, thus causing vasodilation. Because macitentan has high affinity for pulmonary endothelin receptors, it preferentially causes vasodilation in the pulmonary vasculature, thereby decreasing pulmonary vascular pressure. In prospective, randomized controlled trials, macitentan was effective in alleviating symptoms, improving exercise capacity and prolonging the time to clinical worsening in patients with idiopathic PAH. Macitentan was the third endothelin receptor antagonist to be approved in the United States (2013). The current indications are for symptomatic pulmonary arterial hypertension, classified as WHO group 1 (idiopathic). Use of macitentan in forms of PAH due to heart failure, thromboembolic disease or connective tissue disease should be considered experimental as its efficacy in these forms of PAH has not been adequately shown. Macitentan is available in tablets of 10 mg under the brand name Opsumit and the recommended dose is 10 mg once daily. Because of the potential for teratogenicity, macitentan is available for treating women only as a part of a risk evaluation and mitigation strategy (REMS) program in which documentation of adequate methods for birth control are required. Common side effects include headaches, dizziness, anemia, edema, flushing, rhinitis and dyspepsia that are frequent with most vasodilator therapies. Uncommon, but potentially severe adverse reactions include embryo-fetal toxicity, decrease in sperm counts, anemia and hypersensitivity reactions.
Macitentan is associated with a low rate of serum aminotransferase elevations (0% to 4%) that, in clinical trials, was similar to the rate among placebo recipients. These elevations were usually mild, transient and not associated with symptoms, but were above 8 times the ULN in 2% of subjects (vs 0.4% of controls) in at least one long term study. For these reasons, the product label recommends that patients have serum enzymes tested before starting therapy and be alerted to the possibility and symptoms of liver injury during therapy. While there have been no published reports of clinically apparent liver injury with jaundice associated with macitentan, it has had limited general use. Other endothelin receptor antagonists (bosentan, sitaxentan) have been linked to several instances of acute liver injury, some of which have been severe. The onset of illness was usually within 1 to 6 months of starting bosentan and the enzyme pattern was typically hepatocellular or mixed. Immunoallergic features were not present and autoantibodies absent or present in low titer. Macitentan and ambrisentan have not been linked to similar cases.[2]
Macitentan for the treatment of pulmonary arterial hypertension
Macitentan is a dual ET-receptor antagonist (ERA) developed in the setting of extensive efforts to maximize blockade of the ET axis while generating compounds with improved adverse-effect profiles when compared to prior compounds in the class. Unlike bosentan and sitaxsentan, macitentan is not characterized by a sulfonamide structure, but belongs to the sulfamide class of compounds. The presence of a bromine atom in the parent compound of macitentan allowed for straightforward determination of its metabolites. Macitentan is metabolized via oxidative depropylation into ACT-132577 and via oxidative cleavage into ACT-373898. Of the two aforementioned metabolites, only the former, ACT-132577, has been shown to be metabolically active, and like its parent compound, also demonstrates dual-receptor antagonist properties, albeit with less potency. The metabolites are the result of a reaction catalyzed by the cytochrome P450 (CYP), system with enzyme kinetic data showing ACT-132577 primarily formed by CYP3A4 with negligible contribution from CYP2C19. Coadministration of macitentan with the maximum approved dose of ketoconazole, one of the prototypical inhibitors of CYP3A4, resulted in an increase in the concentration of the parent drug, with an area under the curve comparable to the 30 mg dose of macitentan administered during its Phase I evaluation. Nonetheless, it is recommended by the manufacturers that its use be avoided with strong inhibitors of CYP3A4.[3]
At a cellular level, macitentan demonstrates slower dissociation kinetics compared to all other available ERAs, in addition to insurmountable antagonism in functional pulmonary artery smooth-muscle assays. There are multiple lines of evidence showing that heterodimerization of the ETRA and ETRB receptors is necessary for activation, as with other G-coupled proteins, and upon binding macitentan interferes with this heterodimerization, blocking both receptor pathways. It has been demonstrated that macitentan’s functional inhibition constant (KB) ratio of 6.3 is higher than that of both bosentan and ambrisentan (0.7 and 1.0, respectively), indicating significantly more potent antagonism. Both macitentan and its metabolites are highly bound to plasma proteins (>99%), and are primarily excreted via the hepatic and renal systems (combined hepatic and renal excretion of 49.7%±3.9%), with fecal elimination representing 23.9%±4.8% of total excretion. Pharmacokinetic evaluation of the 10 mg dose of macitentan has been shown to be safely tolerated in cirrhosis patients with Child–Pugh classes A, B, and C, with the expected caveat, given its metabolism, of decreased exposure to ACT-132577 in patients with hepatic impairment. Severe renal impairment, defined as a creatinine clearance of 15–29 mL/min, did not have any bearing on the safety profile of macitentan either.
Compared to other available ERAs, it demonstrates superior receptor-binding properties, with consequently improved tissue penetration, and a longer duration of action allowing for once-daily dosing. It has a favorable adverse-effect profile, with notably no demonstrable increase in the risk of hepatotoxicity or peripheral edema, but like other ERAs, it is potentially limited by significant anemia. Phase I data have demonstrated a favorable drug–drug interaction profile and no need for dose adjustment with hepatic and renal impairment. In the pivotal SERAPHIN study, treatment of symptomatic pulmonary arterial hypertension patients with macitentan led to statistically significant improvements in functional class, exercise tolerance, and hemodynamic parameters, in addition to a reduction in morbidity in an event-driven long-term trial.
References
[1] MEGAFINE PHARMA P - WO2016/203489, 2016, A1
[2] LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-. Macitentan. [Updated 2017 Sep 30]
[3] Kholdani CA, Fares WH, Trow TK. Macitentan for the treatment of pulmonary arterial hypertension. Vasc Health Risk Manag. 2014 Nov 25;10:665-73.
Lastest Price from macitentan manufacturers

US $0.00-0.00/KG2025-11-27
- CAS:
- 441798-33-0
- Min. Order:
- 1KG
- Purity:
- 98
- Supply Ability:
- 10000KGS

US $0.00/KG2025-04-21
- CAS:
- 441798-33-0
- Min. Order:
- 1KG
- Purity:
- 99%min
- Supply Ability:
- 100kg